The U.S. Army is designing a new explosive to replace TNT. The new compound forgoes the catchy name in favor of more powerful boom-booms and less toxicity for soldiers.
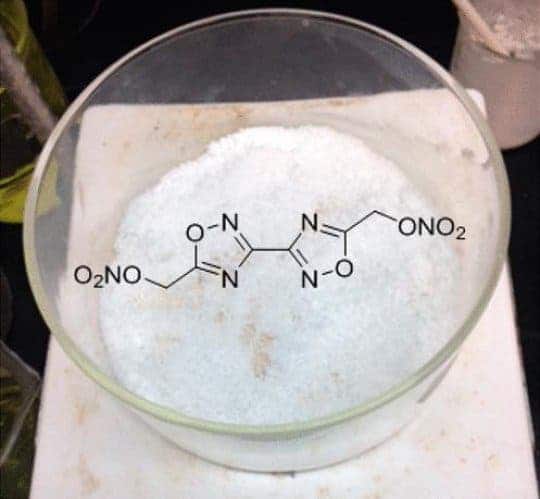
Image credits LANL.
Research at the Los Alamos National Laboratory and the U.S. Army Research Laboratory in Aberdeen, Maryland has resulted in a new “melt-cast” explosive to replace Trinitrotoluene (TNT). The material, bis(1,2,4 oxadiazole)bis(methylene)dinitrate, should be more reactive (i.e. make bigger explosions) than TNT while being less toxic.
More boom for your buck
“The Army and the Laboratory, through the Joint Munitions Program, have been looking for a TNT replacement,” said David Chavez, an explosives chemist at Los Alamos. “Something with non- or low-toxicity that has the right melting point so it can be liquified and cast, for use in a variety of munitions.”
The molecule is a nitrogen-containing compound, which the team refers to as bis-oxadiazole. Chaves used his decade-long experience of developing nitrogen-rich explosive compounds at Los Alamos to develop the material — which has a low explosive sensitivity (won’t go boom when it’s not supposed to) for temperature, pressure, and friction. It also boasts good environmental properties (won’t poison people).
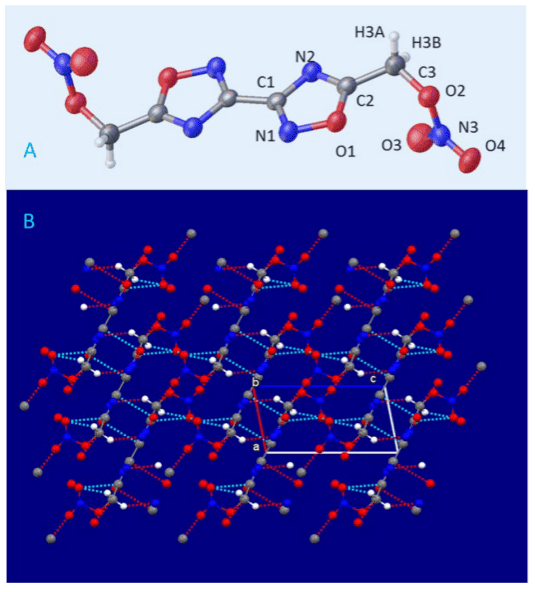
Image credits C. Johnson et al., 2018, OPR&D.
One major challenge the team had to overcome was designing a material that would detonate with more force (aka ‘yield‘) than TNT while being stable enough to melt-cast. The two properties tend to butt heads — stable compounds generally make for poor explosives (fast and energetic reactions).
The team’s earliest attempts only managed a meager 4% yield — far, far too little for an explosive. Several iterations later, Chaves explains, they were up to 44% yield. The final material is a 24-atom molecule packed full of nitrogen, which has 1.5 times the explosive performance of TNT, according to the team.
Another area where bis-oxadiazole should outshine TNT is toxicity, the team writes. The Environmental Protection Agency has listed TNT as a possible carcinogen. Exposure to the material has also been linked to disorders of the blood, such as anemia, and abnormal liver function, according to the Centers for Disease Control.
TNT was first synthesized by Julius Wilbrand, a German chemist, in 1863, to be used as a yellow dye. It took a few years for people to understand that TNT could also explode — up until 1891 — as the material was very stable and not that powerful as an explosive. TNT’s stability, however, made it ideal for pouring into shells and other casings. It became the explosive of choice for ammunition, artillery shells, landmines and all manner of explosive military implements starting in 1901.
Next, Chaves’ team plans to produce a few kilograms of the material to use in explosive testing and toxicity studies.
The paper “Bis(1,2,4-oxadiazole)bis(methylene) Dinitrate: A High-Energy Melt-Castable Explosive and Energetic Propellant Plasticizing Ingredient,” has been published in the journal Organic Process Research & Development.
Was this helpful?



