A huge deposit of lithium along the Nevada–Oregon border could be a game changer for the global battery market. According to a new study, the deposit is so impressive it could change the dynamics of global lithium geopolitics.

Lithium is a soft, silvery-white alkali metal. Under standard conditions, it’s the lightest metal and the lightest solid element. In modern industry, lithium is important as a conductor used in batteries. Lithium batteries have become particularly appealing because of their high energy density. Nowadays, such batteries are used in a range of devices and applications, from smartphones and laptops to electric cars and renewable energy storage.
The increasing demand for electric vehicles and the global shift towards sustainable energy solutions mean that lithium will likely be a crucial resource in the coming decades. However, this also raises concerns about the environmental impact of lithium mining and the geopolitical implications of lithium supply chains. This is why finding a new deposit, and especially one that’s so large, is so important.
According to the new research, the deposit lies in the McDermitt Caldera, a geological formation located on the border between the states of Oregon and Nevada in the United States. A caldera is a large, usually circular depression formed when a volcano erupts and collapses. The McDermitt Caldera is the result of volcanic activity that occurred during the Miocene epoch, roughly 16 million years ago. It’s around 28 miles (45 km) long and 22 miles (35 km) wide.
Volcanic lithium
The McDermitt Caldera is one of several volcanic features in the region, including the Yellowstone hotspot track. The unique geological conditions make McDermitt Caldera an area of particular interest due to its significant lithium deposits. The United States Geological Survey (USGS) and other organizations have shown interest in this region due to the potential of these lithium reserves, which could help reduce dependency on foreign sources of lithium.
But the deposit may be even greater than thought and may have implications not just for the US, but for all countries.
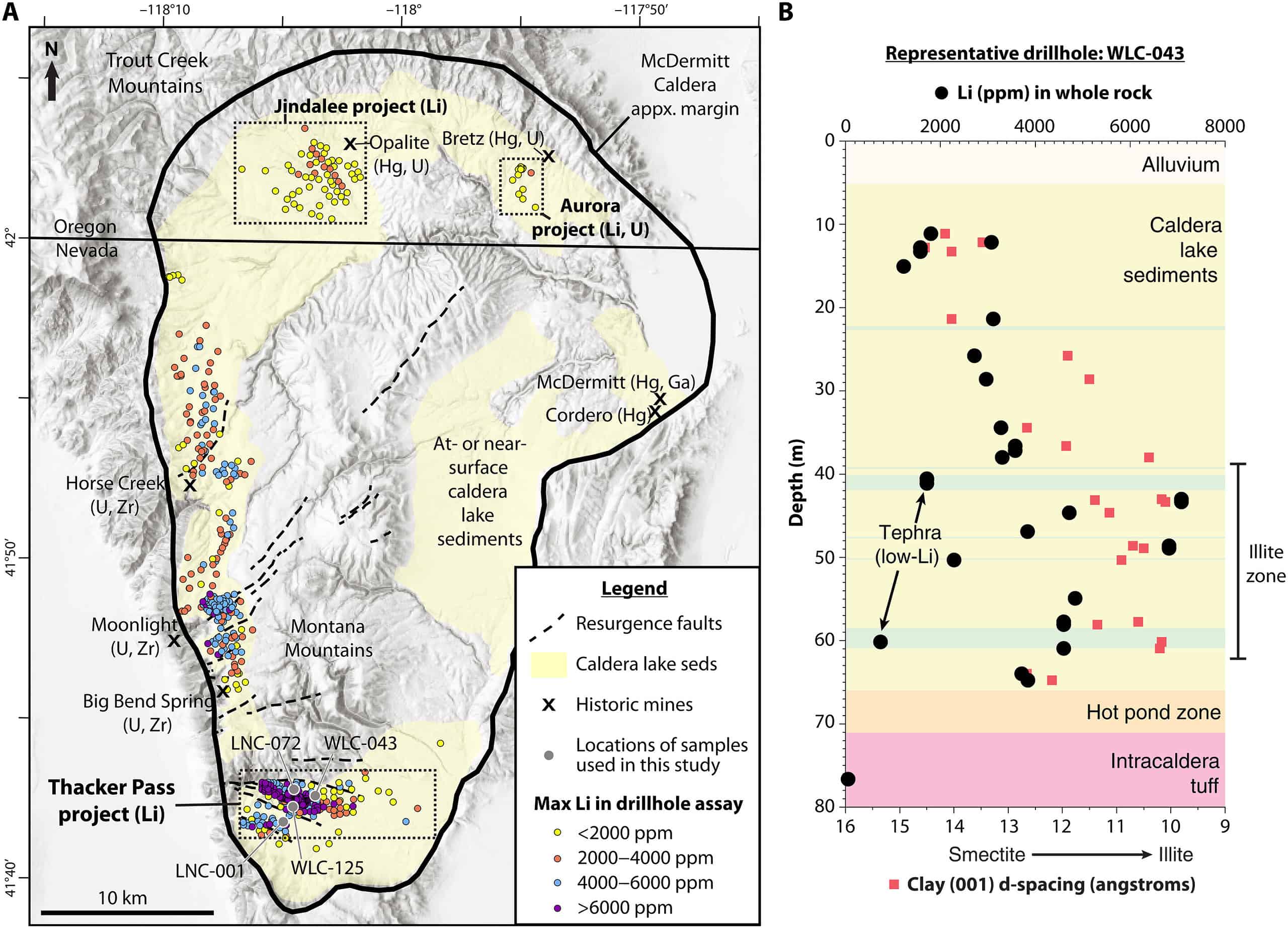
A trio of volcanologists and geologists from Lithium Americas Corporation, GNS Science, and Oregon State University analyzed samples from a clay mineral called illite. This illite contains 1.3% to 2.4% of lithium. This is a very large percentage that’s almost double the lithium in magnesium smectite, a more common lithium-bearing clay mineral. Illite is not as common as magnesium smectite, but it’s richer in lithium. The unique lithium enrichment of illite had not previously been identified.
Researchers also have a theory as to how this happened. They believe that after the volcano erupted and the caldera was formed, a hydrothermal enrichment occurred. In other words, hot, mineralized water (heated by the magma) pushed into the caldera, creating fissures and faults where the water infiltrated and deposited lithium. This process created the lithium-rich illite.

Can we mine it?
Recent calculations estimate an in situ tonnage of 20 to 40 megatons of lithium. Even if this is a ballpark figure and mining the entire caldera may not be economically feasible, it would still make it the biggest lithium deposit on the planet.
“This back-of-the-envelope estimation is calculated using caldera-wide extrapolation of publicly available drill hole data from Lithium Americas Corp. and Jindalee Resources Ltd. and is not a reporting code-compliant mineral resource estimate that considers economic viability,” researchers say.
“Even if this estimation is high due to variations in sediment thickness and/or lithium grade, the lithium inventory contained in McDermitt caldera sediments would still be on par with, if not considerably larger than, the 10.2 megatons of lithium inventory estimated to be contained in brines beneath the Salar de Uyuni in Bolivia, previously considered the largest lithium deposit on Earth.”
Just because a specific resource exists doesn’t mean you can mine it. Lithium is also one of those resources that has the ‘bad habit’ of spreading itself thin, which makes it more expensive to mine.
Typically, there are two ways to mine lithium:
- Brine extraction, which involves extracting lithium from underground reservoirs of saltwater, known as brine. Large salt flats, or salars, contain saline water rich in lithium. The brine is pumped to the surface and stored in large evaporation ponds. Over time (which can be months to years), the water evaporates, leaving behind concentrated salts. These salts are then processed to extract lithium carbonate or lithium chloride. The lithium carbonate can then be further processed to produce lithium metal.
- Hard rock mining, which involves mining lithium-bearing minerals directly from rock formations. The mined ore is then processed to extract the lithium.
Both these methods come with their own environmental challenges. But the McDermitt Caldera opens up a third way for mining lithium — and this way could be more environmentally friendly.
Imagine a volcano erupting and releasing minerals that contain lithium. These volcanic minerals then flow into nearby low-lying areas, like valleys. When these minerals mix and react with the local soil, they create lithium-rich layers. The exciting part about these layers is that extracting them from these sediments might be easier and more environmentally friendly. Unlike other methods, this approach might not need as much energy or complicated processes.
At a place called Thacker Pass, people are optimistic about using this method. They believe it could be a game-changer for reducing the environmental harms of lithium mining. Some recent studies even suggest that if companies look carefully and in the right spots, they could find even more lithium than they expected.
So while probably not all the lithium can be mined, at least some of it can be extracted with relative ease. This being said, there is still plenty of valid opposition to the mining. Thacker Pass (or Peehee Mu’huh in Northern Paiute) sits on land that many local Indigenous groups consider sacred, and Native American activists are fighting a plan to mine lithium in the area.
We need more lithium — but we need to get it sustainably
Thomas Benson, one of the study authors, believes mining can start within 3 years. But having the science part in place is only the first step, ensuring economic and sustainable mining is another.
The discovery of this enormous lithium deposit presents both tremendous opportunities and challenges. While the potential to propel the electric vehicle industry and other sustainable energy applications is evident, there’s a need for responsible and ethical mining practices. These include considering the environmental impacts, ensuring that the rights and concerns of Indigenous groups are respected, and tapping into the deposit in a manner that ensures the sustainability of this invaluable resource.
Moreover, while the deposit’s size is undoubtedly promising, it’s essential to remember that mining is more than just extracting minerals from the ground. It involves complex logistics, infrastructure, and the ability to process these raw materials into usable products. Furthermore, there will be economic, social, and geopolitical ramifications to consider, given that this discovery could potentially shift global supply chains and dynamics.
In essence, the McDermitt Caldera find has paved a path to the future of the battery industry. Whether this path leads to a brighter, more sustainable future or a series of challenges, only time will tell. It will undoubtedly require careful planning, cooperation among different stakeholders, and a steadfast commitment to responsible and ethical practices.






