
Scientists from the University of Sydney employed a fancy quantum computer to dramatically slow down a critical chemical reaction process. The researchers managed to slow down the typically almost instant process by an astonishing factor of 100 billion times, marking a significant advancement in our understanding of molecular dynamics.
“It is by understanding these basic processes inside and between molecules that we can open up a new world of possibilities in materials science, drug design, or solar energy harvesting,” said Vanessa Olaya Agudelo, joint lead author of the new study and a PhD student at the University of Sydney.
“It could also help improve other processes that rely on molecules interacting with light, such as how smog is created or how the ozone layer is damaged.”
Too fast to see
The research team focused on observing the interference pattern of a single atom triggered by a common geometric structure in chemistry known as a ‘conical intersection’. These intersections play a vital role in rapid photochemical processes, including light harvesting in human vision and photosynthesis in plants.
When molecules undergo photochemical reactions, they transition between various electronic states. Conical intersections act as crossroads during these transitions, allowing molecules to switch between different states with remarkable speed. When light hits our eyes we almost instantly process the stimulus as vision, for instance — and it’s all partly due to these conical intersections.
Although chemists have long sought to directly observe these geometric processes since the 1950s, the rapid timescales involved in chemical reactions have posed a significant challenge.
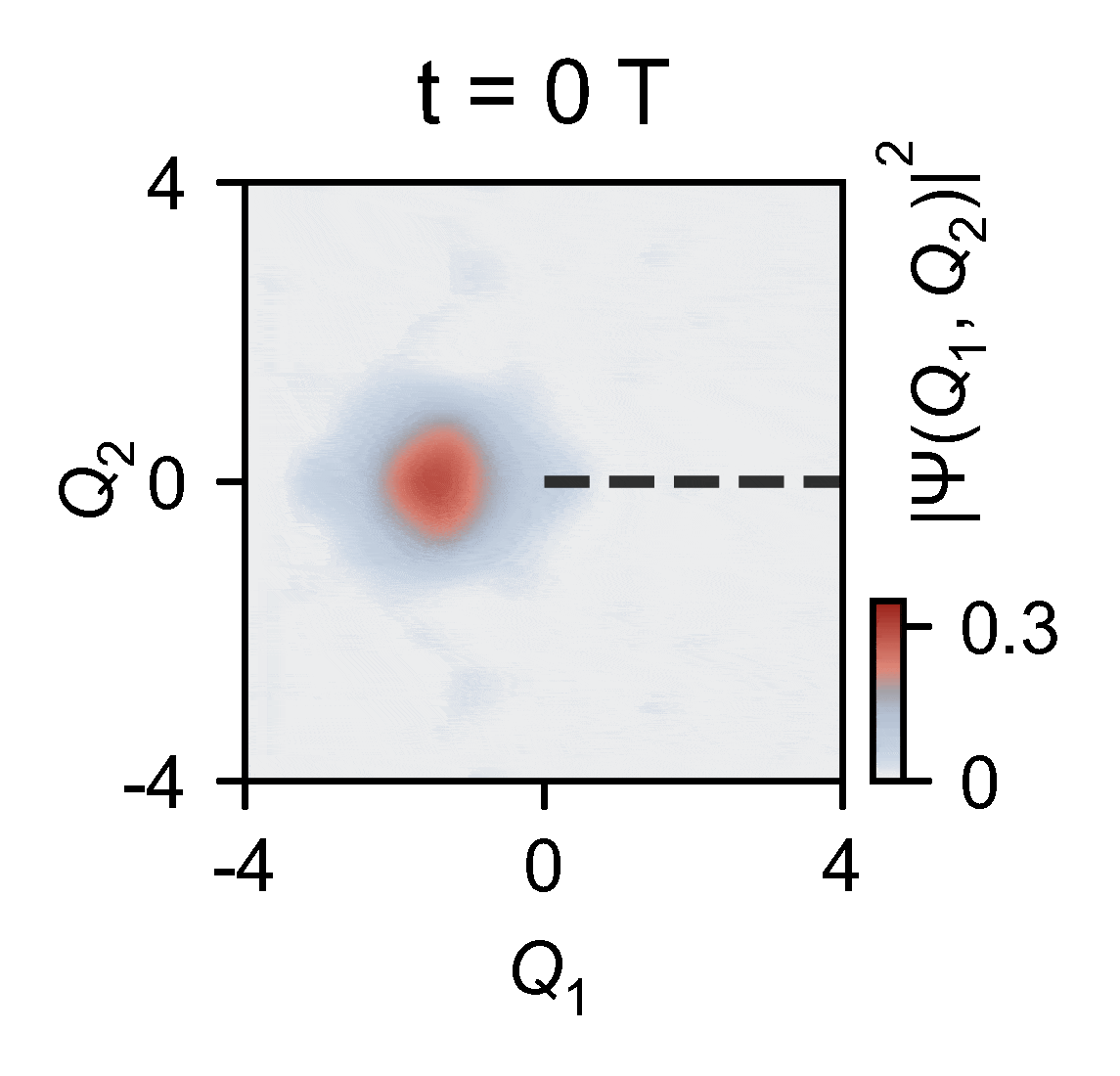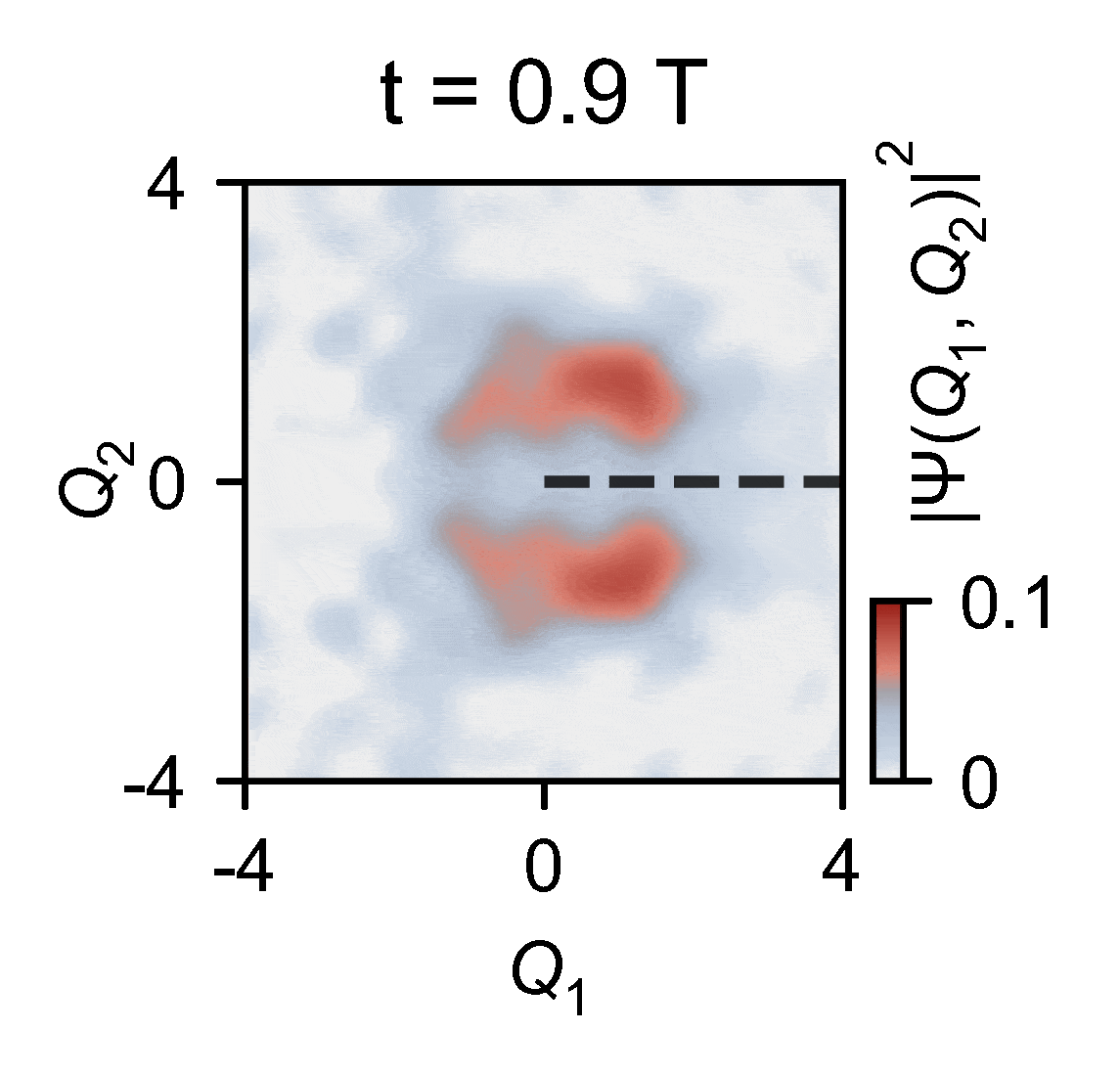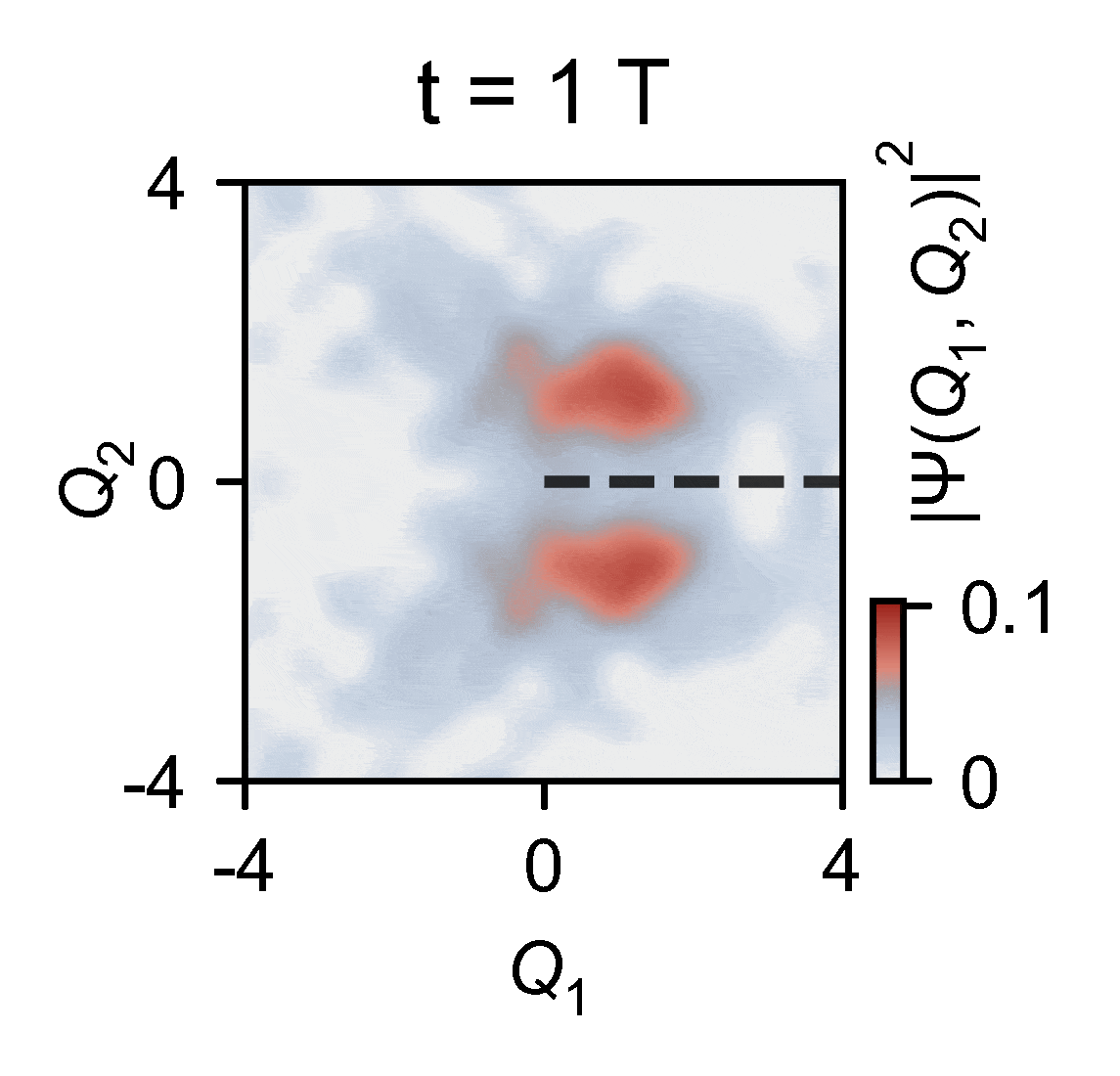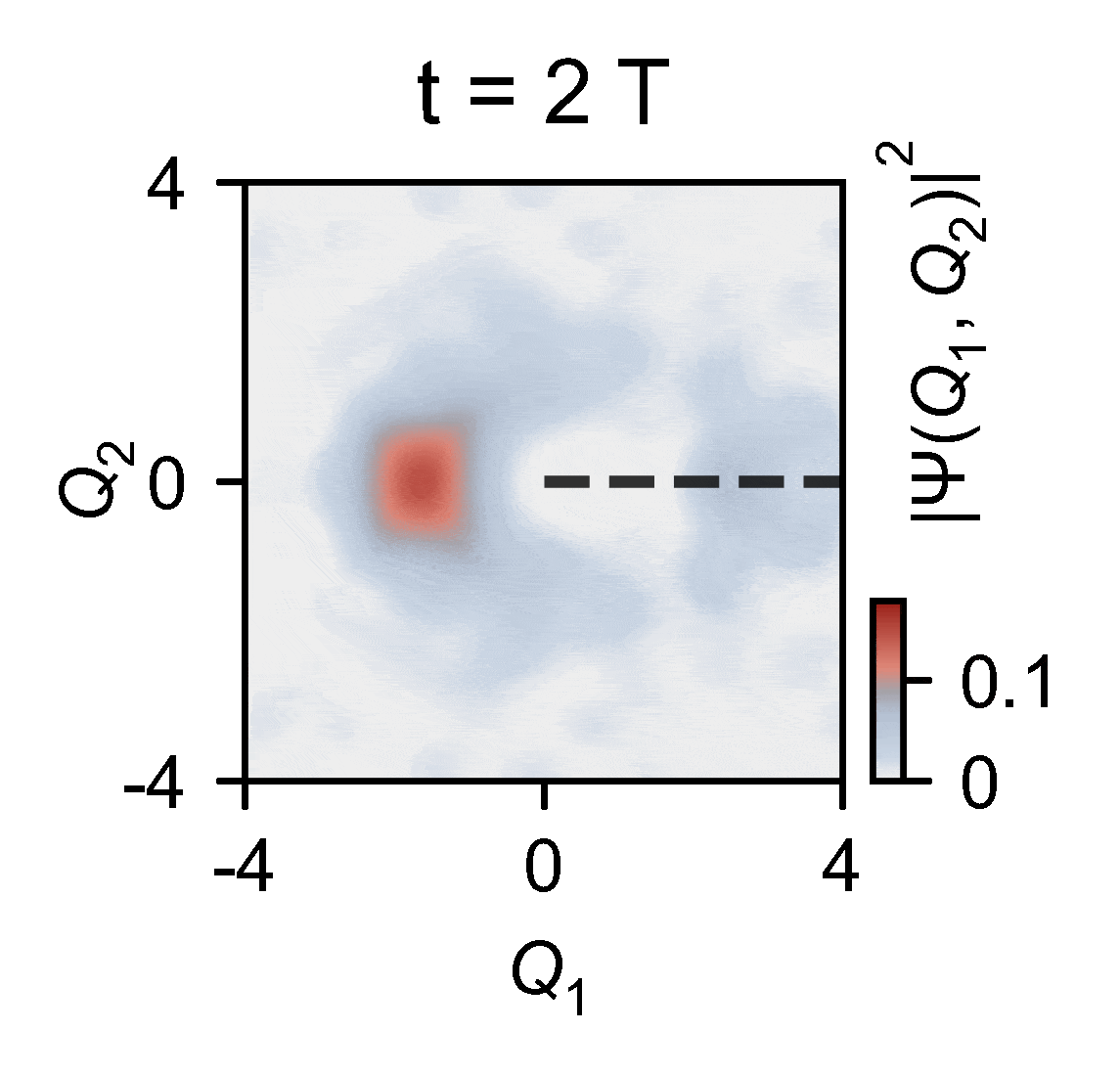
To overcome this obstacle, the quantum researchers devised an ingenious experiment in which they used a quantum computer to reconstruct two-dimensional wavepacket densities of a single trapped ion of ytterbium confined in vacuum by electric fields — a method that enables capturing the intricate behaviors of molecules. This allowed them to map the complex problem onto a relatively compact quantum device and subsequently slow down the simulated process by an extraordinary factor of 100 billion. This is slow enough to see the conical intersections in action.
The outcomes of the experiments harmonize seamlessly with the theoretical model, underscoring the remarkable capability of analog quantum simulators, particularly those actualized using trapped ions. These simulators exhibit remarkable precision in describing the quantum effects arising from the motion of atomic nuclei.
“In nature, the whole process is over within femtoseconds,” said Olaya Agudelo. “That’s a billionth of a millionth — or one quadrillionth — of a second.”
“Using our quantum computer, we built a system that allowed us to slow down the chemical dynamics from femtoseconds to milliseconds. This allowed us to make meaningful observations and measurements.”
A milestone in ultrafast dynamics
In the past, researchers have detected indirect traces of these geometric phases in patterns of scattering and spectroscopic data. But before this breakthrough, scientists had no method at their disposal to directly observe the dynamics of these geometric phases because they simply happened too fast.
Moreover, this isn’t some kind of digital approximation of the process, but rather a direct analog observation of the quantum dynamics of the chemical phenomenon unfolding at a speed that we can observe intermediate steps. Dr. Christophe Valahu, a joint lead author from the School of Physics, drew an analogy to understanding the air patterns around a plane wing in a wind tunnel.
The study’s implications stretch to crucial biological processes like photosynthesis, wherein plants derive energy from sunlight. Molecules involved in photosynthesis transfer energy at incredibly high speeds. But other fields of science where molecules change at fast timescales could also have much to gain.
The findings appeared in the journal Nature Chemistry.






