Ice is one of the most widely studied topics in solid state physics but despite the thousands of academic papers published thus far, there’s so much we don’t know yet. Water can actually freeze in myriad ways, depending on pressure and temperature, to simplify things. On this planet’s surface, water crystallizes in only one way. Whether it’s in an ice cube in your drink or a glacier in the Arctic, it’s all the same ‘hexagonal ice’ or ‘ice on-H’, as it’s known to physicists. But now researchers have for the first time caught water crystallize into ‘ice seven’, an exotic form of ice found naturally in alien environments.

In Kurt Vonnegut’s Cat’s Cradle, a personal favorite, the narrator tracks down the children of Felix Hoenikker, a fictitious Nobel laureate physicist who helped develop the atomic bomb, so he could finish his book about the scientist. Our hero eventually winds up on the Carribean island of San Lorenzo, one of the poorest countries in the world, ruled by a local dictator named “Papa” Monzano. Once on the island, his destiny becomes intertwined with those of the most influential people on the island as the story unfurls into bewilderment. Things happen so fast the narrator eventually becomes the ruler of San Lorenzo. The big plot twist, however, is Papa’s secret: a vial of ‘Ice-Nine’ — a crystalline form of water so stable that in practical terms it would never melt. After Papa died, Ice-Nine was released into the environment, crystallizing all water on Earth, locking it into the Ice-Nine configuration.
“There were no smells. There was no movement. Every step I took made a gravelly squeak in blue-white frost. And every squeak was echoed loudly. The season of locking was over. The Earth was locked up tight.”

Ice Nine actually exists though not at all in the way Vonnegut described in his book. Ice-IX was discovered in 1968 and it exists only under high pressure. Vonnegut’s brother, who held a PhD in physical chemistry from MIT; and published papers on silver iodide and ice formation (cloud seeding), likely served as an inspiration.
Ice-VII (Ice Seven) might not be as intense as the fictitious Ice-Nine but that doesn’t make it any less exciting. Like Ice-Nine, Ice-Seven is stable at room temperature — but you also need liquid water above 3 GPa. Though you can’t find it naturally on Earth, scientists think Ice-Seven is the constituent of giant planets and icy moons.
“These experiments with water are the first of their kind, allowing us to witness a fundamental disorder-to-order transition in one of the most abundant molecules in the universe,” said study lead author Arianna Gleason, a postdoctoral fellow at Los Alamos National Laboratory.
The fastest chiller
Because phase changes can happen faster than the blink of an eye and on the tiny scale of mere atoms, it’s always been a challenge for scientists to capture this moment-to-moment action. “What our new study demonstrates, and which hasn’t been done before, is the ability to see the ice structure form in real time,” said study senior author Wendy Mao, an associate professor of geological sciences and a Stanford Institute for Materials and Energy Sciences (SIMES) principal investigator.
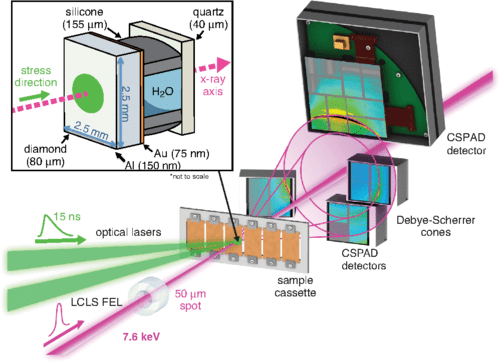
The team overcame the minute timescales involved in the phase transformation thanks to the Linac Coherent Light Source, the world’s most powerful X-ray laser located at the nearby SLAC National Accelerator Laboratory. This machine fires a green-colored laser that vaporizes layers of diamonds on one side of the target (liquid water) producing a rocket-like force that squeezes water to a pressure exceeding 50,000 times that of Earth’s atmosphere at sea level.
A separate laser beam is fired just as the water is compacted from another instrument called the X-ray Free Electron Laser. The beam is composed of very short, bright pulses that last only a femtosecond or quadrillionth of a second. It’s this strobing X-ray laser that eventually recorded the crystallization of water on a molecule-by-molecule basis, like a flip book. According to the paper published in Physical Review Letters, the phase change took only six nanoseconds during which the water molecules bonded into rod shapes. Previously, some physicists suggested the molecules bonded into spheres.
The same experimental setup could be used to explore water crystallization under a myriad of pressure and temperature scenarios. Specifically, in the case of Ice-Seven, the new-found knowledge will help scientists come up with better models of alien environments like Jupiter’s moon Europa but also remote events such as comet impacts.
“Any icy satellite or planetary interior is intimately connected to the object’s surface,” Gleason said. “Learning about these icy interiors will help us understand how the worlds in our solar system formed and how at least one of them, so far as we know, came to have all the necessary characteristics for life.”
Was this helpful?



