All the building blocks of life, formed by chains of carbon, hydrogen, nitrogen and oxygen, came from outer space. How exactly organic compounds brought by asteroids or comets reacted to form amino acids, carbohydrates, fats and proteins is still a mystery, but what better place to look for answers than the very origin of life: deep space.
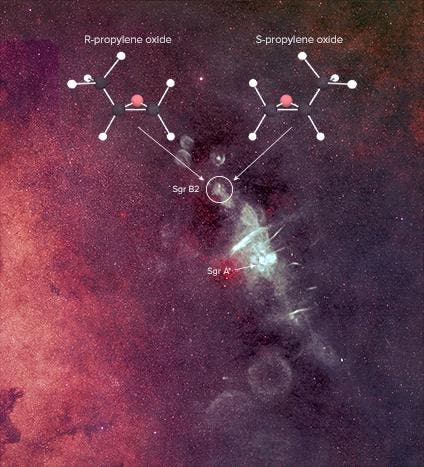
Some of our efforts have paid off. In a star-forming cloud, thousands of light-years away called Sagittarius B2, astronomers have found an organic element for the very first time whose properties are thought to be essential to forming life.
This molecule is called propylene oxide (CH3CHOCH2), and it’s the first molecule we’ve found outside Earth that exhibits chirality.
Chirality is a simple but essential idea to modern chemistry that describes two mirror forms. It’s easier if you imagine the chirality of a molecule by looking at your hands. We have a left and right hand, which both seem to look, feel and function the same. Were you to transplant your left hand in the place of your right hand, though, you’d be in a bit of trouble. You’d literally have two left hands and be extremely clumsy at working things with two hands.
The idea is that the left and right hand are mirror images, but not identical. Likewise, any molecule that differs from its reflection is called chiral. Otherwise it’s called achiral.
Chirality is paramount in chemistry and its effects can be very far reaching. Although we’re talking about the same molecule, the ‘left’ one could react very differently from the ‘right’ one.
A famous case that’s often used as a textbook example for chirality is the story of Thalidomide, a sedative drug used in the late ’50s and early ’60s. This drug was chiral and sold in a 50/50 mix of left and right. While the left molecule acted as a sedative, the right one impeded the proper growth of the foetus when taken in the first trimester by pregnant women.
We will never know the extent of the damage this tragedy caused, but considering this was a drug marketed in 46 countries, hundreds if not thousands of babies were born with abnormalities because of it. A hard lesson learned about chirality. Another, much less serious, example of chirality is Aspartame which is a sweetening agent hundreds of times sweeter than sucrose. But it’s mirror image is bitter!
Chirality also underpins all the building blocks of life, like sugars or proteins. For instance, all organisms use the right-hand form of the sugar ribose which forms the backbone of DNA and gives it its signature double-helix twist. If you dig deeper, and biologists have, you’ll notice that all essential building blocks for life are either left-handed or right-handed, but never present in both forms. This is homochirality, and many scientists are left scratching their heads when they try to explain how peculiar pattern emerged.
The findings in Sagittarius B2 might serve us well, though. Propylene oxide, an organic chemical used to make plastics here on Earth, was detected because every molecule a signature radio wave which was picked up by two radio telescopes on Earth, one in West Virginia, U.S., the other in Australia.
“This is the first molecule detected in interstellar space that has the property of chirality, making it a pioneering leap forward in our understanding of how prebiotic molecules are made in the universe and the effects they may have on the origins of life,” Brett McGuire, a Jansky postdoctoral fellow with the National Radio Astronomy Observatory (NRAO) in Charlottesville, Virginia, and author of the study published in the journal Science.
“Propylene oxide is among the most complex and structurally intricate molecules detected so far in space. Detecting this molecule opens the door for further experiments determining how and where molecular handedness emerges and why one form may be slightly more abundant than the other,” added Brandon Carroll, a chemistry graduate student at the California Institute of Technology in Pasadena and co-author of the paper.
Determining whether the deep space propylene oxide is left or right handed is very difficult because their spectral lines are identical, but we know from tests in the lab that this molecule is chiral for sure. This is what McGuire and Carroll need to determine next and it’s a very big challenge, to say the least.
If the researchers can determine from thousands of light-years away that some propylene oxide molecules are left or right handed, their distribution (say one is far more common than the other) might glean clues to the origin of homochirality here on Earth.





