Swedish researchers were looking to create artificial bone, but in doing so they may have come across something more important. By analyzing the process of their bone imitation, the researchers were able to study how our bones grow at an atomic level, morphing from an unstructured mass into a perfectly arranged structure.
It’s a well-established fact that our bones grow in stages, but what exactly goes on in each of these stages had previously been a mystery. Martin Andersson, Professor in Materials Chemistry at Chalmers University of Technology, Sweden, along with colleagues, developed a method for creating artificial bone using 3-D printing. Once this technology is fully developed, the team hopes to create nature-mimicking implants that might replace metal and plastic implants currently in use.
However, while Andersson and colleagues were printing bones, they couldn’t help but notice that their process was extremely similar to the environment that living tissue grows in. Using electron microscopes, the team studied how material turned from an amorphous mush into an orderly structure that resembles bone, all at the atomic level.
“A wonderful thing with this project is that it demonstrates how applied and fundamental research go hand in hand. Our project was originally focused on the creation of an artificial biomaterial, but the material turned out to be a great tool to study bone building processes. We first imitated nature, by creating an artificial copy. Then, we used that copy to go back and study nature,” Andersson said in a statement.
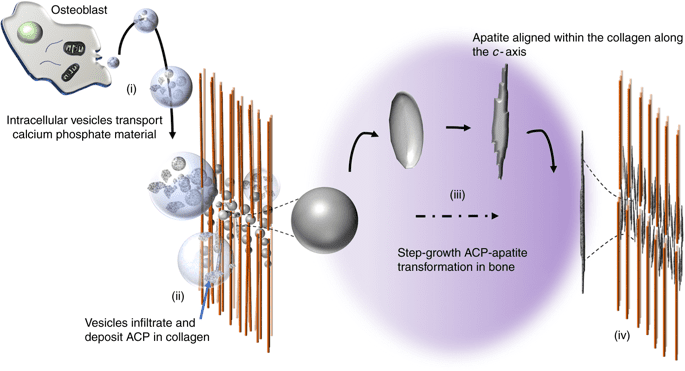
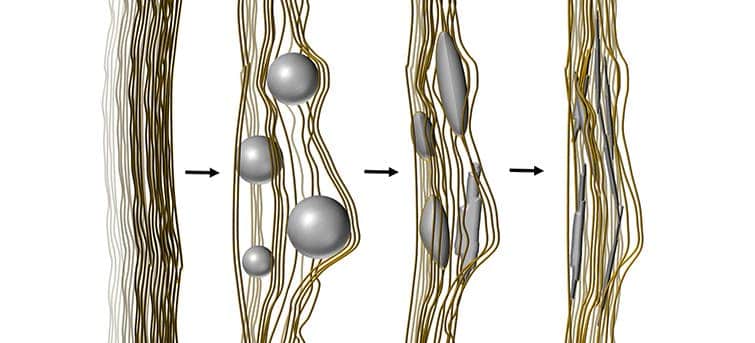
Writing in the journal Nature Communications, the Swedish researchers explain how bone mineralization first starts with strings of the protein collagen — the smallest structural building block in the skeleton. Cells then send spherical particles called vesicles to the site, where they bind between the collagen strings. There, the vesicles, which are made of calcium phosphate, transform from an amorphous mass into an ordered crystalline structure.
The crystallization process is not biological but rather purely physical, following the laws of thermodynamics. The calcium phosphate molecules are drawn to where the energy level is the lowest, and therefore where there is a more stable state.
These findings could prove highly useful, not only in manufacturing nature-mimicking bones for implants but also for treating bone diseases.
“Our results could be significant for the treatment of bone disease such as osteoporosis, which today is a common illness, especially among older women. Osteoporosis is when there is an imbalance between how fast bones break down and are being reformed, which are natural processes in the body,” said Martin Andersson.
Drugs that seek to address this imbalance may be improved thanks to this study’s newfound knowledge, the authors conclude.






