New research at RMIT University is looking into liquid metals as a solution to drug-resistant bacteria.
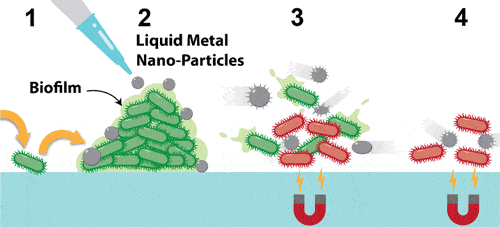
The approach the team is working on involves using magnetic particles of liquid metals to physically destroy bacteria, side-stepping the use of antibiotics entirely. The study describes how this technique can be used to destroy both bacteria and bacterial biofilms — protective, layered structures that house bacteria — without harming human cells.
A shred of hope
“We’re heading to a post-antibiotic future, where common bacterial infections, minor injuries and routine surgeries could once again become deadly,” says Dr Aaron Elbourne, a Postdoctoral Fellow in the Nanobiotechnology Laboratory at RMIT, and the paper’s lead author.
“It’s not enough to reduce antibiotic use, we need to completely rethink how we fight bacterial infections.”
The rising levels of antibiotic resistance recorded throughout the world is a very scary development, one that we’ll have to tackle sooner rather than later. Modern antibiotics fundamentally changed the rules of life for us when they were first developed 90 years ago. Before that, any infection was basically the luck of the draw: even a routine medical intervention or the most unassuming of wounds could become infected, and even the humblest infection could kill.
They still can, but modern antibiotics offer us a level of protection that people in the past could only pray for. Still, overuse and misuse of these compounds are forcing pathogens to adapt and survive, and they’re doing so much faster than we can develop new, more powerful drugs. It’s estimated that antibiotic-resistant bacteria cause in excess of 700,000 deaths per year, a figure which could reach 10 million a year by 2050 (which would make it deadlier than cancer). Bacteria’s ability to form biofilms further complicates the matter, as such structures render them virtually immune to all existing antibiotics.
Antibiotics are chemical compounds that prevent bacteria from functioning properly. They can do this through a range of methods: by blocking their ability to form proteins, by breaking down their membrane, or by interfering with their ability to reproduce. Human cells and bacterial cells are similar but different enough that antibiotics can be made to target the latter and leave the former unaffected.
The team wanted to develop a whole new method to attack pathogens, one that does away completely with chemical means (which bacteria can adapt to).
“Bacteria are incredibly adaptable and over time they develop defences to the chemicals used in antibiotics, but they have no way of dealing with a physical attack,” Dr. Elbourne explains.
“Our method uses precision-engineered liquid metals to physically rip bacteria to shreds and smash through the biofilm where bacteria live and multiply.”
The team’s approach involves the use of nano-sized droplets of liquid metal. When exposed to a low-intensity magnetic field, these droplets change shape and grow sharp edges.
To check how effective they would be at the task, the team placed droplets in contact with a bacterial biofilm then made them change shape. The sharp edges broke down the biofilm and physically ruptured the bacterial cells inside, the team found. They proved effective against both Gram-positive and Gram-negative bacterial biofilms. After 90 minutes of exposure to the particles, both biofilms were destroyed and 99% of the bacteria inside were killed, the team explains, suggesting that they would be effective as a wide-range treatment option. Human cells were left unaffected by the nanoparticles.
The team says that their method is versatile enough to be used in multiple settings and approaches. For example, a coating of the nanoparticles could be sprayed on implants to help prevent infections for hip or knee replacements. They also plan to explore its effectiveness against fungal infections, cancerous tumors, and build-ups such as cholesterol plaques.
“There’s also potential to develop this into an injectable treatment that could be used at the site of infection,” said Dr Vi Khanh, a Postdoctoral Research Fellow at the North Carolina State University and co-author of the paper.
The nanoparticles are currently undergoing preclinical trials in animals. If all goes well, human trials could start in a few years.
The paper “Antibacterial Liquid Metals: Biofilm Treatment via Magnetic Activation” has been published in the journal ACS Nano.



