As the world shifts towards clean energy, lithium has become one of the most sought-after minerals on the planet. This light metal is a crucial component in the batteries that power electric vehicles and store energy from renewable sources. But while lithium may be a green energy solution, the process of mining it has raised environmental and ethical concerns. Where does lithium come from and how is it produced? Here’s an explainer with everything you should know, including the environmental impacts.

Lithium is a highly reactive alkali metal with excellent heat and electrical conductivity. Such characteristics make it especially useful to manufacture lubricants, pharmaceuticals, glass and, most importantly, the cathode for lithium-ion batteries used to power electric cars and consumer electronics.
But the soft, silver-white metal can’t be found in its elemental form, as it’s highly reactive. Instead, it’s present as a constituent of salts or other compounds. Most of the lithium available in the market can be found as lithium carbonate, a more stable compound that can then be transformed into other chemicals or salts.
Lithium salts can be found in underground deposits of clay, mineral ore and brine, as well as in geothermal water and seawater. Most of the world’s lithium comes from mines, from where it’s extracted. However, briny lakes, also known as salars, have the highest concentration of lithium, ranging from 1,000 to 3,000 parts per million.
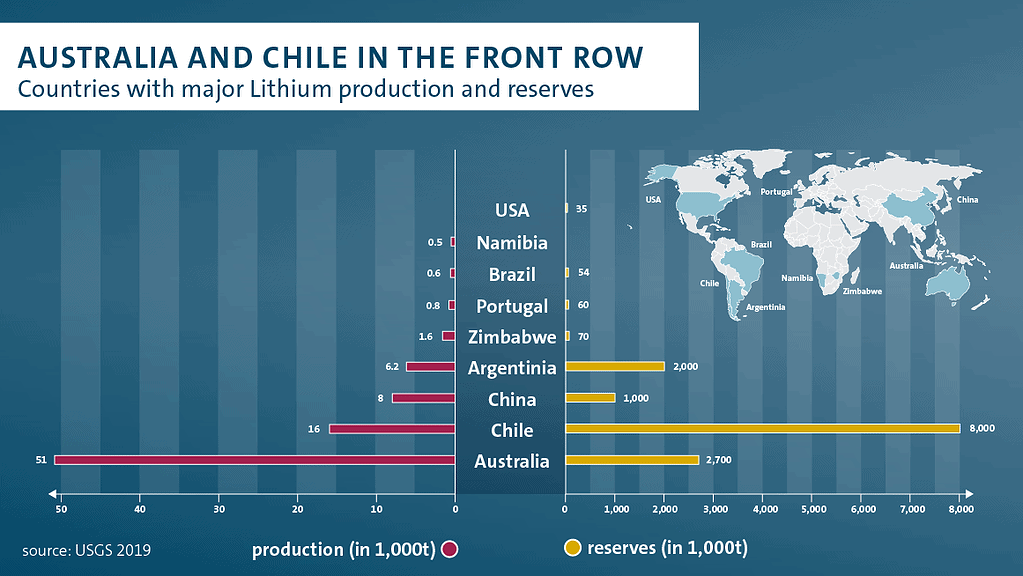
Lithium can be found in a variety of geological settings, but the largest reserves are in lithium-rich brine deposits in Chile, Argentina, and China. Other significant reserves are found in spodumene pegmatites, which are a type of lithium-bearing rock, in Australia and North America.
Lithium extraction

The salars with the highest lithium concentrations are located in Bolivia, Argentina, and Chile, in an area called “the lithium triangle.” Lithium obtained from salars is then recovered in the form of lithium carbonate.
Brine extraction in salars is normally a very long and cumbersome process that can take from eight months to three years. Mining starts by drilling a hole and pumping brine to the surface. The unearthed minerals are left outside to evaporate for months, first creating a mix of manganese, potassium, borax, and salts which is filtered and placed into another evaporation pool.

It will take between 12 and 18 months for that mix to be filtered enough in order to be able to extract the lithium carbonate, also known as “white gold”. However, lithium carbonate fields, or ponds, don’t always look white. Seen from above, lithium ponds range in color from a pinky white, to turquoise, to a highly concentrated, canary yellow. While it’s cheap and effective, the process needs a lot of water, estimated at 500,000 gallons per ton of lithium extracted.
This creates a lot of pressure on local communities living in nearby areas. For example, in Chile’s Salar de Atacama, mining has caused the region to lose 65% of the region’s freshwater, putting a huge strain on local farmers, who rely on agriculture and cattle for their livelihoods and now need to source their water from somewhere else.
How much lithium is there in the world?
The global lithium reserve base is estimated to be around 39 million metric tons, according to the USGS. Chile is the country with the largest lithium reserves, with an estimated 7.5 million metric tons, followed by Australia with 6.3 million metric tons, and Argentina with 3.4 million metric tons. Other countries with significant reserves include China, the United States, and Russia.
Lithium is a relatively rare element, making up only 0.0017% of the Earth’s crust. However, it is found in a variety of geological settings, including lithium-rich brine deposits and spodumene pegmatites.
Lithium-rich brine deposits are the most important source of lithium, accounting for around 60% of global reserves. Spodumene pegmatites are another important source of lithium, accounting for around 30% of global reserves. These are a type of lithium-bearing rock that is found mainly in Australia and North America. Spodumene mining involves extracting the rock, crushing it, and then processing it to extract the lithium.
Lithium can also be found in other sources such as lithium-bearing clay deposits, lithium-bearing ores, geothermal fluids, and lithium-bearing mineral springs.
The increasing demand for lithium in the production of batteries for electric vehicles and energy storage systems has led to a surge in exploration and development activity. As a result, new lithium resources are being identified and developed around the world, which is likely to increase the global reserve base.
But is there enough lithium to satisfy our requirements for transitioning towards a 100% renewable future? The currently known reserves of lithium are more than enough to meet the projected demand for the year 2100. In 2021 lithium extraction peaked at an industry record of 100,000 metric tons, so there is still a lot more lithium to mine.
The environmental impact of lithium mining
Lack of water in the region is not just the single potential problem with lithium mining. Toxic chemicals can leak from the evaporation pools to the water supply, such as hydrochloric acid, which is used in the processing of lithium – as well as other waste products that can filter out of the brine.
In the United States, Canada, and Australia, lithium is usually extracted from the rock by using more traditional methods. Nevertheless, this still requires the use of chemicals in order to extract it in a useful form. In Nevada, researchers found evidence of pollution in fish swimming 150 miles downstream from a lithium processing operation, for example.
Another concern is the discharge of saline water back into the environment. The process of extracting lithium from brine generates large volumes of highly saline water, which can harm local ecosystems if not properly managed.
A report by Friends of the Earth argued that extracting lithium can affect the soil and causes air contamination. In the area Salar del Hombre Muerto in Argentina, residents complain that lithium polluted streams that are used by humans and livestock, while in Chile there were clashes between mining firms and locals.
In some cases, mining companies have been accused of forcibly displacing communities and violating the rights of indigenous peoples. In Chile, for example, there have been reports of mining companies abusively buying land from farmers without their consent.
In addition, the rapid expansion of lithium mining in some regions has led to a boom-and-bust cycle, with local economies becoming dependent on the mining industry and then suffering when mining activity slows down.
Improved technologies for lithium extraction
Researchers argue that there’s a need to develop new extraction technologies so that the industry might manufacture batteries in a more environmentally friendly way. That’s why across the world many are looking for new alternatives, such as battery chemistries that replace cobalt and lithium with more common and less toxic materials.
Nevertheless, some of these experimental batteries that are less energy-dense or more expensive could still end up having a negative effect on the environment. “A less durable, yet more sustainable device could entail a larger carbon footprint once you factor in transportation and the extra packaging required,” said Christina Valimaki an analyst at Elsevier.
Being able to recycle lithium-ion plays a key role as well. In Australia, research showed that only 2% of the country’s 3,300 tons of lithium-ion waste was recycled. That can cause problems, as unwanted electronics with batteries still inside can end up in landfills, where toxic metals and ionic fluids can leak into underground water reservoirs.
The Birmingham Energy Institute is using robotics technology initially develop for nuclear power plants to look for ways to remove and dismantle potentially explosive lithium-ion cells from electric vehicles. There were a number of fires at recycling plants where lithium-ion batteries have been stored improperly.
A key problem is that manufacturers are usually secretive regarding what actually goes into the batteries, which makes it difficult to recycle them properly. Today, recovered cells are mostly shredded, leading to a mixture of metals that can be separated using pyrometallurgical techniques.
Growing demand

The global enchantment over mobile devices and all kinds of technological gadgets has led to a growing demand for lithium-ion batteries. That’s especially applicable to electric vehicles, as the world seeks to stop using fossil fuels in the near future to reduce global greenhouse gas emissions. The battery of a Tesla Model S, for example, uses around 12 kg of lithium, thousands of times more than the lithium found in a consumer device.
By 2025, lithium demand is expected to increase to approximately 1.5 million metric tons of LCE (lithium carbonate equivalent) and over 3 million tons by 2030, mainly spurred by the auto industry. For example, Volkswagen hopes to launch more than 70 electric car models in the next 10 years.
To get an idea of how much the battery industry, and by extension, lithium mining, has grown, consider the fact that battery tonnage has grown by an order of magnitude in eight years, from 70GWh to 700GWh.
For now, the demand for lithium far outpaced production, which is reflected in the metal’s price. According to Benchmark Mineral Intelligence, its lithium price index had risen by 182.6% year-on-year as of mid-December 2022. These prices could stay high for some time as demand is forecast to remain very high.
The open question is the consequences that such demand will have on the environment and the communities near the salt mines where the lithium is extracted. The more gadgets and electric vehicles, the more lithium that will be needed in the future, raising the need to develop more environmentally friendly extraction techniques.






