A team of German researchers has developed a new way to deal with seasonal and avian influenza viruses. Their approach involves wrapping the pathogens in chemically-modified bacteriophage capsids, rendering them unable to infect human cells.
The team hopes their work will help usher in new treatment options against such viruses. The method was tested in the lab with very encouraging results and is currently under investigation for possible applications against the coronavirus.
Viral straightjacket
“Pre-clinical trials show that we are able to render harmless both seasonal influenza viruses and avian flu viruses with our chemically modified phage shell,” explained Professor Dr. Christian Hackenberger, Head of the Department Chemical Biology at the Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) and Leibniz Humboldt Professor for Chemical Biology at HU Berlin. “It is a major success that offers entirely new perspectives for the development of innovative antiviral drugs.”
Current antiviral treatments only attack the influenza virus after it has infected our cells, the team reports, which is certainly useful — but preventing infection in the first place would be much more desirable and effective.
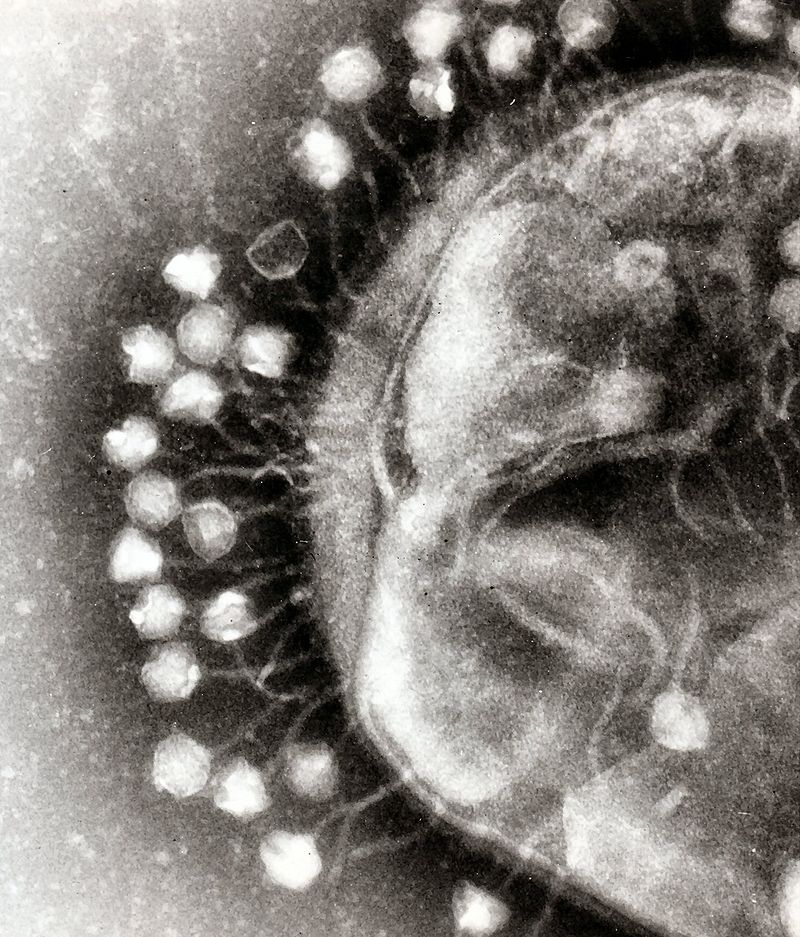
The trials — which used infected human lung tissue samples — showed that perfectly fitting a phage capsid onto these viruses can be used to neutralize their ability to infect lung cells. The capsid was specially developed by the team for this job, and works by binding itself to all the (hemagglutinin) proteins the virus can use to gain access through the membranes of human cells. During the infection process, these proteins bind to sugar molecules sprinkled through the membrane of lung tissue cells to allow entry. The core mechanism of this process, however, relies on the virus creating multiple bonds with a cell, rather than a single one.
Their quest to develop an inhibitor for these proteins started six years ago. The plan was to make such an inhibitor functionally resemble the membrane of a human lung cell. The team’s quest led them to the Q-beta phage, a harmless species of bacteriophage that lives in our intestines and usually preys on E.coli. The team removed and attached ligands (binders) to its casing — sugar molecules in this case — to act as bait binding sites for the virus’ proteins
“Our multivalent scaffold molecule is not infectious, and comprises 180 identical proteins that are spaced out exactly as the trivalent receptors of the hemagglutinin on the surface of the virus,” explained Dr. Daniel Lauster, a former Ph.D. student in the Group of Molecular Biophysics (HU) and now a postdoc at Freie Universität Berlin. “It therefore has the ideal starting conditions to deceive the influenza virus — or, to be more precise, to attach to it with a perfect spatial fit. In other words, we use a phage virus to disable the influenza virus!”
When samples of tissue infected with flu viruses were treated with the phage capsid, the influenza viruses were practically unable to reproduce. High-resolution cryo-electron microscopy and standard cryo-electron microscopy revealed that the modified capsids completely cover the viruses.
While definitely encouraging, the findings call for more preclinical studies to assess the method’s viability and safety for human use. We don’t yet know, for example, if the capsids themselves would elicit an immune response in mammals, and if such a response would enhance or impair their effect. And, of course, it has yet to be proven that the inhibitor is also effective in humans.
For now, the team is content to know that their approach has great potential and that it is “the first achievement of its kind in multivalency research,” according to Professor Hackenberger. The approach, he adds, is biodegradable, non-toxic, doesn’t cause an immune response in cell cultures, and is, at least in principle, applicable to other viruses and possibly even bacteria. The team is currently focusing on adapting it to the SARS-CoV-2 virus.
The paper “Phage capsid nanoparticles with defined ligand arrangement block influenza virus entry” has been published in the journal Nature Nanotechnology.
Was this helpful?



