One team of researchers trying to peer into the body took inspiration from submarine flicks and created a novel E. coli strain whose gas-filled protein sacks have the ability to reflect sound waves. They plan to use the bacteria to determine if treatments are making it to the desired spots in the body and working properly.
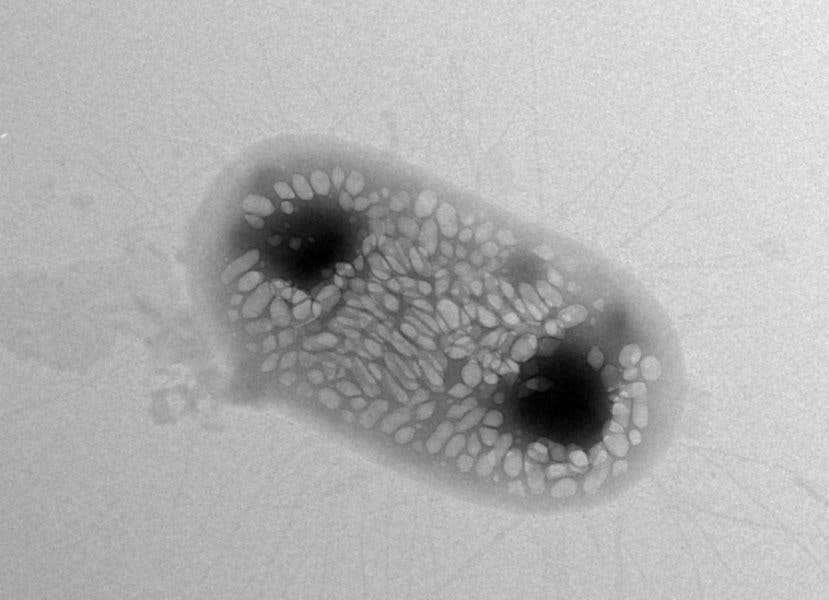
Image credits Anupama Lakshmanan / Caltech.
Sometimes in medicine, we humans are frustratingly big compared to what we’re trying to heal. That makes is really hard to snoop around and see if the medicine we’re using gets to the right place and doing its job. It also doesn’t help that we’re quite opaque.
Hear here
One team of researchers is trying to work around both of those issues by making bacteria do the ‘seeing’ for us — using sonar. Their genetically-engineered strand of E. coli carries gas vesicles which can reflect incoming sound waves, in a process similar to how submarines reflect signals from sonar. The end goal is to inject such bacteria into a patient’s body to help fight disease — by targeting tumors, for example — and then using ultrasounds to determine their location. Such a technique would allow doctors to tell if treatments are making it to the right place in the body and whether or not they’re working as intended.
It’s not the first time anyone has considered using bacteria to fight illness. These little bits of life have shown great efficiency when treating disorders such as irritable bowel disease, and there is evidence suggesting they can be drafted to destroy cancer cells. However, the paper is the first to describe a reliable way of monitoring and drawing useful information from the bacteria while they’re deployed.
“We want to be able to ask the bacteria, ‘Where are you and how are you doing?'” says corresponding author Mikhail Shapiro.
“The first step is to learn to visualize and locate the cells, and the next step is to communicate with them.”
Similar approaches in the past have relied on light, mainly by ‘tagging’ cells with a “reporter gene” that encodes a fluorescent protein. However, such strategies have the decided disadvantage of only being usable in tissue samples removed from the body — because light can’t pass very deep through tissues.

Image credits New Savanna.
Shapiro’s team worked around this issue by substituting light for ultrasounds. Sounds — mechanical vibration — can pass through the body with relative grace compared to light. But that also poses an issue: sound can just as easily pass through bacteria.
Insulated prokaryotes
Shapiro says he had an epiphany six years ago, when he first learned about water-dwelling bacteria that regulate their buoyancy using gas-sacks (vesicles). These structures, he realized, could be used to bounce back sound waves enough to make the bacteria distinguishable from other types of cells (since gas is a much poorer carrier of sound than the surrounding tissues). The team proved that Shapiro’s approach works with ultrasounds — the things doctors use to look at babies in the womb — at least in the guts of mice.
Next, they had to move the genes encoding these vesicles from the original bacteria into something they could use medicinally. Their pick was Escherichia coli, which is commonly used in microbial therapeutics such as probiotics.
“We wanted to teach the E. coli bacteria to make the gas vesicles themselves,” says Shapiro. “I’ve been wanting to do this ever since we realized the potential of gas vesicles, but we hit some roadblocks along the way. When we finally got the system to work, we were ecstatic.”
Transferring the genetic blueprints into E. coli proved trickier than expected. The team first tried copy-pasting the genes isolated from a water-dwelling bacterium called Anabaena flos-aquae, but this failed to produce any gas-vesicles in the receiving bacteria. So they switched tactics and grafted the genes from Bacillus megaterium, a bacterium more closely related to E. coli. That didn’t work either.
Finally, the team mixed genes from both species, and this time it worked. The authors explain that the genes which encode the gas vesicles act like either bricks or cranes for the structures. Some of the proteins they encode are the actual building blocks, while others just help to glue everything together.
“Essentially, we figured out that we need the bricks from Anabaena flos-aquae and the cranes from Bacillus megaterium in order for the E. coli to be able to make gas vesicles,” says first author Raymond Bourdeau.
Further lab tests with mice showed that the engineered strain of E. coli could also be picked up using ultrasounds. The addition of the gas-vesicles doesn’t affect the bacteria’s viability. Being the “first acoustic reporter gene for use in ultrasound imaging,” it will still take a few years for the technology to be vetted enough for use in humans. However, the team is confident that it will “soon” be available for scientists who work with animals.
“We hope it will ultimately do for ultrasound what green fluorescent protein has done for light-based imaging techniques,” says Shapiro, “which is to really revolutionize the imaging of cells in ways there were not possible before.”
The paper “Acoustic reporter genes for noninvasive imaging of microbes in mammalian hosts,” has been published in the journal Nature.
Was this helpful?



