For the first time, astronomers have detected primordial oxygen gassing out from a comet. ESA’s Rosetta shuttle made the surprising in situ discovery on the comet 67P/Churyumov–Gerasimenko. The fact that they found pure oxygen molecules (O2) indicates that the oxygen came from the initial comet formation.

Oxygen is the third most abundant element in the Universe, but its simplest molecular form (O2) is surprisingly hard to find and pinpoint. Even in star forming clouds, oxygen is highly reactive so you generally see it bound to other elements, which is why it was quite surprising to find it on a comet.
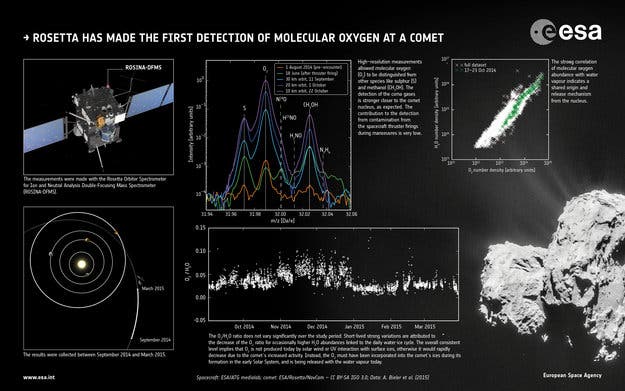
“We weren’t really expecting to detect O2 at the comet – and in such high abundance – because it is so chemically reactive, so it was quite a surprise,” says Kathrin Altwegg of the University of Bern, and principal investigator of the Rosetta Orbiter Spectrometer for Ion and Neutral Analysis instrument, ROSINA.
Rosetta has been studying 67P/Churyumov–Gerasimenko for over a year and has detected an abundance of different gases pouring from its nucleus. Water vapour, carbon monoxide and carbon dioxide are found in abundance, with nitrogen, sulphur and even noble gases also reported. We know this first hand, as Rosetta’s Philae lander successfully made the first soft landing on a comet nucleus when it touched down on Comet Churyumov–Gerasimenko on 12 November 2014.
“It’s also unanticipated because there aren’t very many examples of the detection of interstellar O2. And thus, even though it must have been incorporated into the comet during its formation, this is not so easily explained by current Solar System formation models.”
Overall, the team analyzed 3000 samples collected around the comet in the past year and found an abundance of 1–10% relative to H2O, with an average value of 3.80 ± 0.85% – 10 times more than astronomers were expecting. No ozone was detected.
Ref: “Abundant molecular oxygen in the coma of 67P/Churyumov–Gerasimenko,” by A. Bieler et al is published in the 29 October 2015 issue of the journal Nature.






