Vanderbilt researchers have developed a new sensor which makes brain cells light up like fireflies.
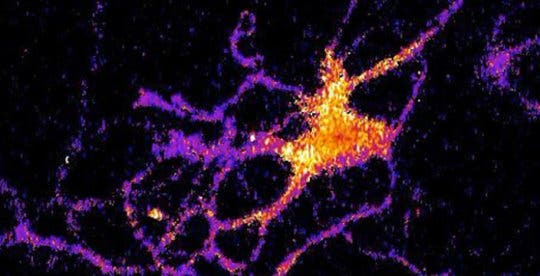
Credit: Johnson Lab / Vanderbilt University
The probe relies on a modified version of luciferase, the class of oxidative enzymes that produce bioluminescence (not to be confused with photoproteins). In nature, several types of organisms use luciferase to regulate their light emission, the most famous being fireflies. Several types of mushrooms and marine creatures also use the same enzyme, though, lighting up through luminescence, not fluorescence.
With this in mind, researchers wanted to see if they can also use it to make brain cells light up, which would have a number of potential medical applications. It’s not the first time something like this has been attempted, but usually, these efforts rely on fluorescence which has some drawbacks.
“For a long time neuroscientists relied on electrical techniques for recording the activity of neurons. These are very good at monitoring individual neurons but are limited to small numbers of neurons. The new wave is to use optical techniques to record the activity of hundreds of neurons at the same time,” said Carl Johnson, Stevenson Professor of Biological Sciences, who headed the effort.
“Most of the efforts in optical recording use fluorescence, but this requires a strong external light source which can cause the tissue to heat up and can interfere with some biological processes, particularly those that are light sensitive,” he said.
To put it simply, fluorescence requires light to function and it interacts with this light, causing unwanted interference. Luminescence, on the other hand, works completely in the dark. Johnson and his collaborators — Associate Professor Donna Webb, Research Assistant Professor Shuqun Shi, post-doctoral student Jie Yang, biological sciences doctoral student Derrick Cumberbatch, Professor Danny Winder, and molecular physiology and biophysics postdoctoral student Samuel Centanni — genetically modified a type of luciferase to light up when exposed to calcium ions. To insert this into the brain cells, they used a creative method, piggybacking on a virus that attaches to the neurons. Calcium was used here because calcium levels spike when neurons receive signals, so it’s a substance tied to natural neural activity.
They’ve successfully tested the method and showed it works, but it’s still a crude version. In future research, scientists want to determine the exact sensitivity of the method and see how they can finesse the delivery.
“We’ve shown that the approach works,” Johnson said. “Now we have to determine how sensitive it is. We have some indications that it is sensitive enough to detect the firing of individual neurons, but we have to run more tests to determine if it actually has this capability.”
Journal Reference: Jie Yang, Derrick Cumberbatch, Samuel Centanni, Shu-qun Shi, Danny Winder, Donna Webb, Carl Hirschie Johnson. Coupling optogenetic stimulation with NanoLuc-based luminescence (BRET) Ca sensing. Nature Communications, 2016; 7: 13268 DOI: 10.1038/ncomms13268






