
In an attempt to further out understanding of cell deformation and mechanics, researchers at the Technische Universität München (TUM) have built an artificial cellular model that can change its shape and move on its own.
Cells are complex biological objects that exhibit sophisticated metabolic functions. Primordial cells, however, were much simpler in design: just a membrane and a couple of molecules. Mimicking this simple, minimalist design, German researchers built a cell-like model with a biomechanical function that allows it to move and change its shape without external influence.

The model is made of a membrane shell, two different kinds of biomolecules and, of course, a fuel. The envelope, which in a biological cell corresponds to the vesicle, is made of a double-layered lipid membrane, just like in a natural cell. The membrane was filled with microtubules, like those made by cellular cytoskeletons and kinesins. These tiny proteins act like motors and move objects along the cytoskeletons of real cells. During experiments, these motors constantly push tubules alongside each other sparking movement. The kinetic energy is fueled by adenosine triphosphate (ATP).
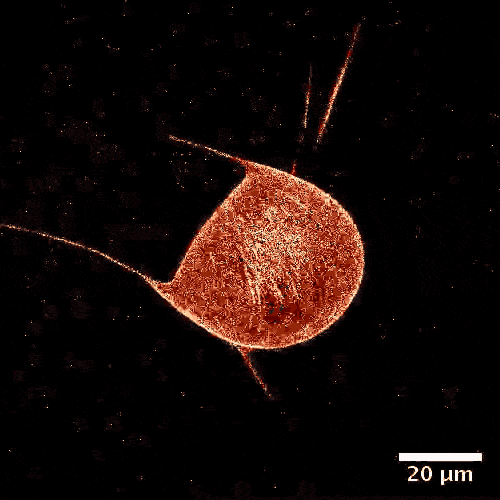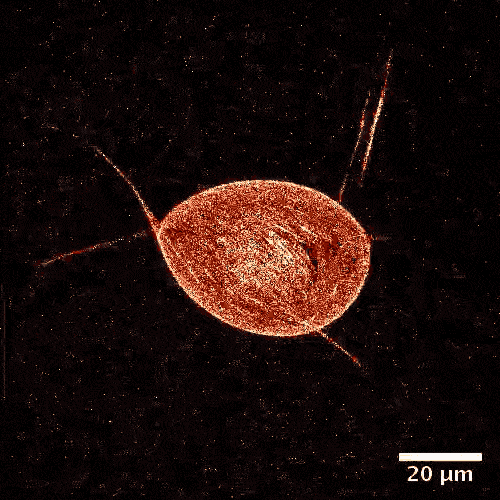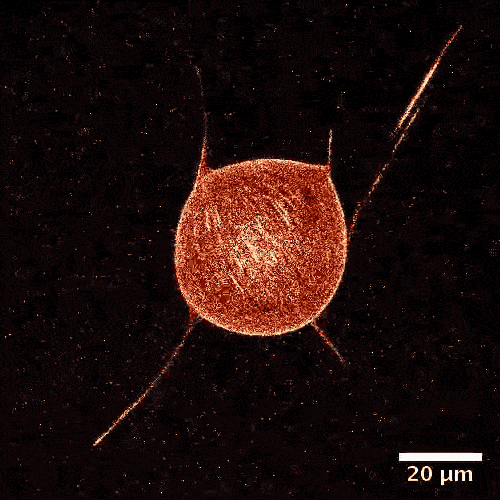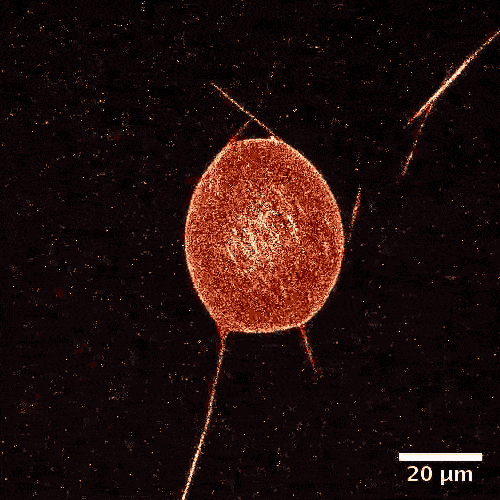
From a physical perspective, the microtubules form a two-dimensional liquid crystal under the membrane, which is in a permanent state of motion.
“One can picture the liquid crystal layer as tree logs drifting on the surface of a lake,” explains Felix Keber, lead author of the study. “When it becomes too congested, they line up in parallel but can still drift alongside each other.”
“With our synthetic biomolecular model we have created a novel option for developing minimal cell models,” explains TUM-Prof. Andreas Bausch. “It is ideally suited to increasing the complexity in a modular fashion in order to reconstruct cellular processes like cell migration or cell division in a controlled manner. That the artificially created system can be comprehensively described from a physical perspective gives us hope that in the next steps we will also be able to uncover the basic principles behind the manifold cell deformations.”
Findings appeared in the journal Science.
Was this helpful?



