Renewable energy and electric vehicles need not only high density storage mediums to become viable, but ones that can be recharged fast as well. Scientists have now developed a new battery, very similar to the popular lithium-ion variety used to power your smartphone, that charges in under a minute and still works perfectly after 7000 cycles. Moreover, the battery is based on aluminum making it both easier and cheaper to manufacture.

Because aluminium is the most abundant metal in the Earth’s crust, many labs have attempted to use it’s ions on the cathode side of batteries instead of lithium, which is mined in a select few parts of the world. Efforts so far have been plagued by numerous difficulties, though. Discharge voltages have been as low as 0.55 V and the cathodes disintegrated very fast, causing the battery to lose 85% of its lifetime in only 100 cycles.
A team at Stanford recently unveiled a prototype that dismisses most of these shortfalls. Remarkably, their breakthrough came more or less by accident when the researchers were investigating metal oxide films doped with graphite to increase their conductivity. But these films also showed battery-like behavior. Hongjie Dai and colleagues eventually built an aluminium-ion battery, where aluminium chloride ions (AlCl4–) are stored at the cathode when the battery is charged. On discharge, the ions migrate to the anode where they combine with metallic aluminium to form Al2Cl7–. When the researchers investigated the graphite cathode’s chemistry they discovered that AlCl4– anions reversibly intercalate between the graphite layers.
The first versions had some serious flaws, though. Because the AlCl4– ions are so bulky, the maximum charge and discharge rates were significantly hampered by the natural graphite plates. This challenge was solved by making a custom graphite foam by depositing carbon onto nickel and then dissolving the metal in acid to produce a carbon structure with large atomic spaces that allowed the AlCl4– ions in and out rapidly with little or no damage.
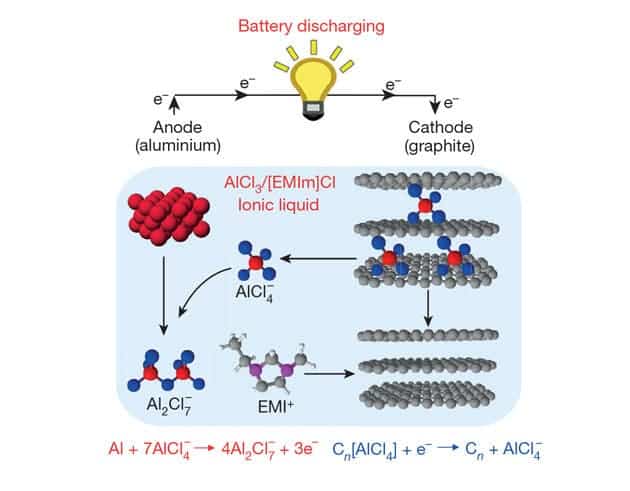
The foam and an ultra-dry electrolyte help the battery discharge at around 2V, while granting an energy capacity on par with lead acid and nickel–metal hydride batteries. The prototype lost little of its energy carrying capacity even after 7000 cycles. In contrast, lithium-ion batteries start to fade away after 1000 cycles. Perhaps most remarkably, the battery can safely be completely recharged in less than 60 seconds or 100 times faster than a lithium-ion battery, as reported in Nature.
With all this in mind, the team believes their work provides an interesting contender for rechargeable batteries in home appliances or storing electricity for the grid or microgrids. At present, the energy density is limited by the bulky AlCl4– ions, but this is being improved.
‘This is the first step to demonstrate that reversible cycling of aluminium is possible,’ says Electrochemist Don Sadoway of the Massachusetts Institute of Technology, US,. ‘The barrier to entry for lithium-ion in automobiles is not the energy density – at 150W/h/kg that’s plenty already. The barrier is cost: lithium-ion is far too costly and will remain too costly. Most people in the battery world are working on the wrong problem and these people deserve credit for addressing the right problem.’
Was this helpful?



