In the US alone, over 30 million people have toxoplasmosis. The parasite that causes the disease, Toxoplasma gondii, is commonly passed through cat feces. But while toxoplasmosis can be problematic in some cases, it has no detectable symptoms for most people.
Now, a group of researchers believes they know why — and it involves brain cells sacrificing themselves to sound the alarm.
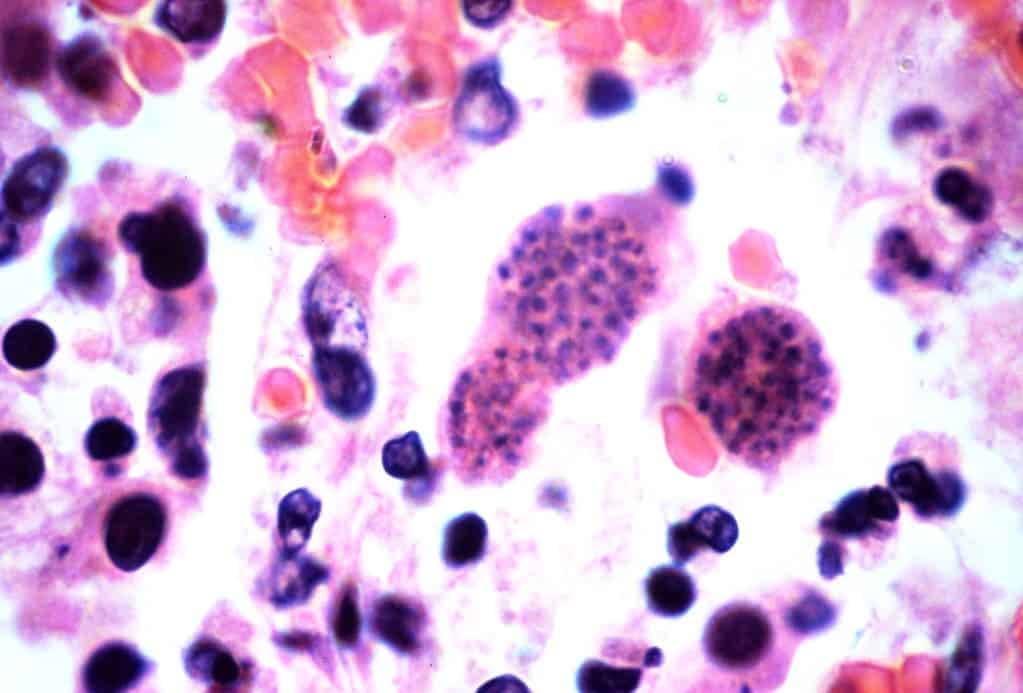
According to a new study, the toxoplasmosis parasite is kept in check by immune cells called microglia. These are the resident defenders of the brain, acting as macrophages and operating as the main form of immune defense of the central nervous system.
These microglia also release a unique molecule, called IL-1α (also called hematopoietin 1) which can serve many roles, but in this case, it’s used as an alarm system. The microglia must sacrifice themselves to secrete IL-1α to let other cells know that something is wrong, a finding that could have implications for many conditions that affect the brain, from stroke to MS or neurodegenerative diseases.
For decades, conventional wisdom claimed that the brain was disconnected from the immune system, but recent research has shown that this is not the case. Microglia are at the center of this debate, but it’s not easy to study them — until recently, lab equipment struggled to distinguish between brain microglia and other types of immune cells. Now, Samantha Batista from University of Virginia used an elegant approach to study these microglia.
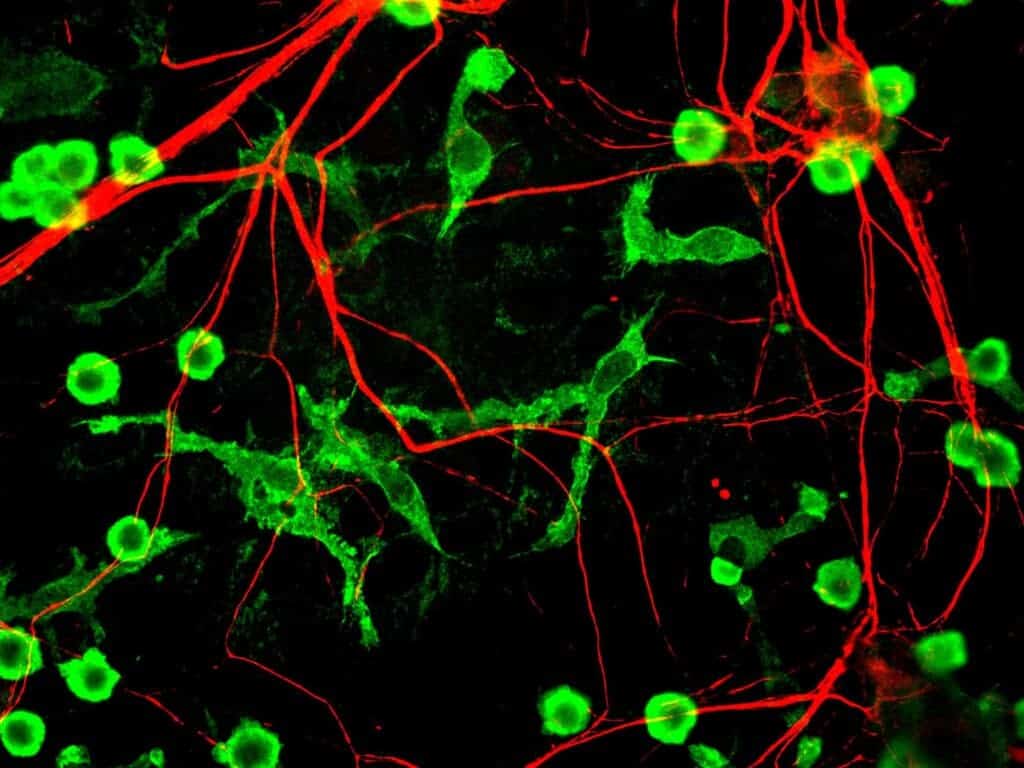
She and her colleagues found that infection causes microglia to burst in an inflammatory fashion — a process that other immune cells don’t undergo. They used the approach to study how microglia control the Toxoplasma gondii infection. People with a healthy immune system typically have no problem in keeping the infection in check and exhibit no symptoms at all. However, immunocompromised people can become very sick.
This is interesting for a number of conditions
“Understanding pathways like this could be beneficial for other diseases involving neuroinflammation,” Batista said. “We can ask whether promoting this pathway is helpful in situations where you need more of an immune presence in the brain, such as infections or cancers, and also whether inhibiting this molecule could be helpful in diseases driven by too much neuroinflammation, like multiple sclerosis. Targeting one specific pathway like this one could have less off-target effects than targeting inflammation more broadly.”
Now that researchers know how the microglia sound the alarm, they are now looking at how they detect a threat in the first place.
“The immune system must enter the brain to fight dangerous infections,” said Harris, who is part of UVA’s Carter Immunology Center. “We now understand how microglia sound the alarm to protect the brain. We suspect that similar signals are missed or misinterpreted in Alzheimer’s disease, opening up an exciting new research avenue in the lab.”
The study was published in Nature Communications.
Was this helpful?



