British researchers have demonstrated three ways gold nanotubes can be used against cancer: 1) high resolution in-vivo imaging; 2) drug delivery vehicles; 3) agents that destroy cancer itself. Their work shouldn’t be viewed as yet “another” hack that seeks to eradicate cancer. We need to be more realistic than this. Instead, the findings have the potential to be a great measure that both diagnoses and treats cancer at the same time, complementing conventional surgery and, hopefully, avoiding the need for chemotherapy.
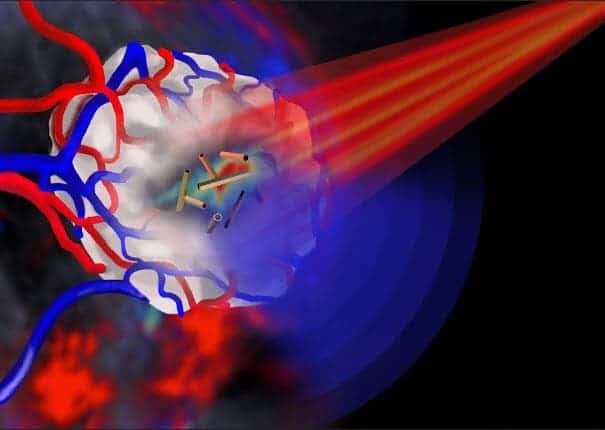

The scientists at the University of Leeds made the first successful demonstration of the biomedical use of gold nanotubes in a mouse model of human cancer. They injected the gold nanotubes intravenously then shone a pulsed infrared laser beam. By adjusting the brightness of the laser pulse, the researchers were able to control whether the gold nanotubes were in imaging mode or cancer-destruction mode. In addition, beforehand, the researchers claim the nanotubes can have their lengths adjusted in order to absorb a precise wavelength.
[SEE] How gold is made and how it got to our planet
In image-mode, the gold nanotubes absorbed the the energy from the laser pulse and generated ultrasound. Using multispectral optoacoustic tomography (MSOT), researchers then read the ultrasound waves and detected the gold nanotubes.
For cancer destruction, there were two options:
- Use a stronger laser beam to rapidly raise the temperature in the vicinity of the nanotubes so that the temperature was high enough to destroy cancer cells.
- Load the central hollow core of the nanotubes with a therapeutic payload.
Although gold is not particularly harmful to the body (unless you pass a certain threshold), the team coated the nanotubes with a protective sodium polystyrenesulfonate (PSS) coating. Ultimately, the nanotubes are safely excreted from the body, after they meet their purpose.
“High recurrence rates of tumors after surgical removal remain a formidable challenge in cancer therapy. Gold nanotubes have the potential to enhance the efficacy of these conventional treatments by integrating diagnosis and therapy in one single system,” said lead author Sunjie Ye, who is based in the School of Physics and Astronomy and the Leeds Institute for Biomedical and Clinical Sciences at the University of Leeds.
Findings appeared in Advanced Functional Materials






