It sounds like a plot from Frankenstein, but apparently there’s no limit to how versatile flatworms can be. Previously, researchers at Tufts University determined that the small, yellow worm can retain its memories after it head was severed. As a reminder, flatworms can regrew new heads following decapitation. Now, the same team yet again guillotined some flatworms and interrupted gap junctions, which are protein channels that enable cells to communicate with each other by passing electrical signals back and forth – just to see what would happen. Yes, the flatworm grew a new head, but it was that of another flatworm species. They eventually induced the same flatworm species to grow the heads and brains of multiple other, closely related species. There’s a lot of biology and behaviour encoded in genes, but these findings show that tweaking physiological mechanisms in a live body can actually cause new anatomical developments. We might have uncovered a new form of epigenetics.
“It is commonly thought that the sequence and structure of chromatin – material that makes up chromosomes – determine the shape of an organism, but these results show that the function of physiological networks can override the species-specific default anatomy,” says the paper’s senior and corresponding author Michael Levin, Ph.D., who holds the Vannevar Bush Chair in biology and directs the Center for Regenerative and Developmental Biology in the School of Arts and Sciences at Tufts. “By modulating the connectivity of cells via electrical synapses, we were able to derive head morphology and brain patterning belonging to a completely different species from an animal with a normal genome.”
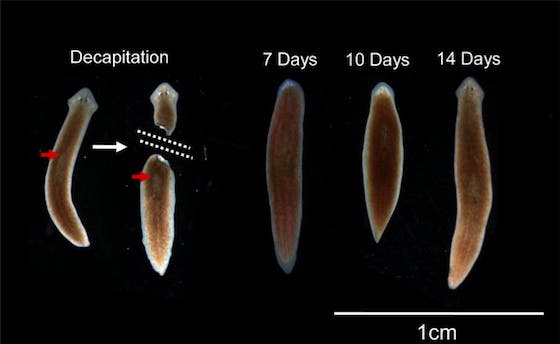
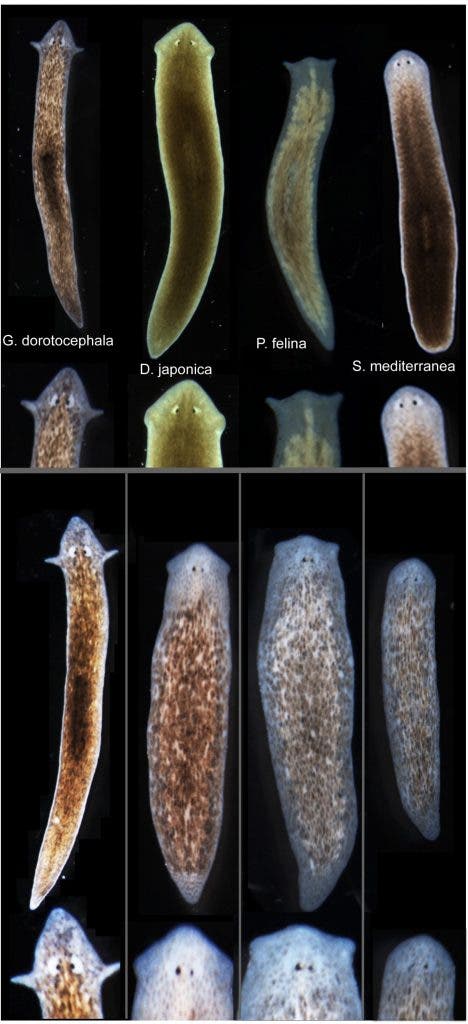
The researchers at Tufts worked with Girardia dorotocephala – a flatworm species known for its extraordinary regenerative capability. After severing the head, the researchers introduced a transient perturbation of physiological connectivity among cells. Despite the worm had the same genome, once it started to regenerate a new head its morphology, and consequently that of the brain, was altered. By varying this disturbance the researchers grew heads that resemble those of flatworm cousins. The closer the two species were related, the easier it was to effect the change, suggesting there’s an evolutionary connection that allowed these worms to morph their body parts. You can see from the picture posted above just how insanely close the worms grew their heads similar to other species.
Oddly enough, this change is temporary. Weeks after the planaria completed regeneration to the other species’ head shapes, the worms once again began remodeling and re-acquired their original head morphology.
The flatworm is really an inexhaustible well of wisdom. For instance, they’re the primary object of study for researchers working the field of longevity. Some claim that flatworms are immortal. The flatworm not only is able to regenerate its old, dead cells, but it can literary grow a new brain, gut or tail when severed in two. Both cut ends grow into a new individual. Over the course of their several year long research, Notthingham University scientists have cloned a few thousand individuals starting from one single flatworm that was cut in two, which were also at their own term cut in two, and so on so forth. What can we learn from the flatworm’s latest trick? Well, doctors might find a way to fix birth defects or cause new biological structures to grow after an injury, according to Levin. “These findings raise significant questions about how genes and bioelectric networks interact to build complex body structures,” he says.