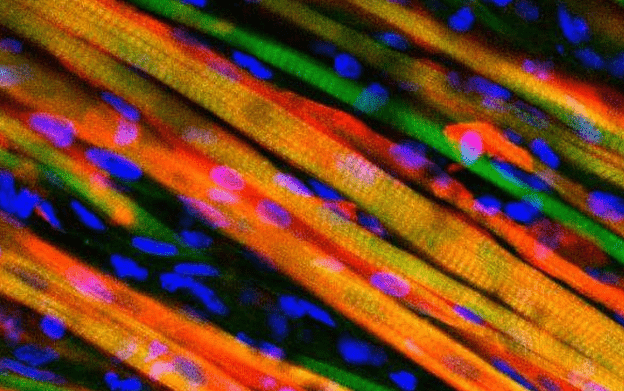
Heralded as one of the biggest advances in the field, scientists at Duke University have engineered muscle tissue that is up to ten times stronger than anything previously achieved. The muscle can contract similarly to native neonatal skeleton muscle and, most importantly, it demonstrates self-healing ability – again, just like the real thing. To demonstrate their work, the researchers also implanted the muscles in bionic mice and followed the muscle fibers as they grew through a window on the back of the living animal.
“The muscle we have made represents an important advance for the field,” said Nenad Bursac, associate professor of biomedical engineering at Duke. “It’s the first time engineered muscle has been created that contracts as strongly as native neonatal skeletal muscle.”
Artificially creating muscles is a great challenge, but if muscle implants are demonstrated in humans, then a slew of injuries or degenerative muscle diseases could be addressed therapeutically. The team led by Bursac found that the two most important aspects that need to be considered when engineering muscles are the contractile muscle fibers and a pool of muscle stem cells, known as satellite cells.
[ALSO READ] Synthetic muscle made from nylon is 100 times stronger than human muscle
Self-healing muscle grown in a lab
The latter is of significant important in all muscle carrying organisms. Your muscles has many, many such satellite cells layered around them, waiting in standby until they’re efforts are required. For instance, when an injury occurs to the muscle, like those following a car accident or even a hefty workout, the satellite cells activate and begin the regeneration process.
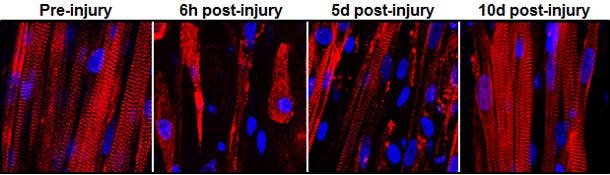
The key to the team’s success was successfully creating the microenvironments—called niches—where these stem cells await their call to duty.
“Simply implanting satellite cells or less-developed muscle doesn’t work as well,” said graduate student Mark Juhas. “The well-developed muscle we made provides niches for satellite cells to live in, and, when needed, to restore the robust musculature and its function.”
First, the researchers ran a series of trials on the muscles in the lab, submitting it to electrical impulses. The muscle contracted showing strength 10 times greater than anything demonstrated in a lab previously. Then, the muscle was damaged using snake venom and satellite cells activated, proving their environment can help the muscle regenerate.
A window peering into progress
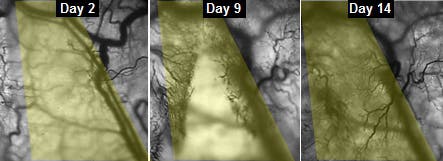
Then followed the trial on mice. The researchers inserted their lab-grown muscle into a chamber in the back of mice and implanted a window to follow progress. The evolution turned out to be spectacular, as the muscles proved to grow stronger as the days and weeks passed by. To make things easier, the muscle was genetically engineered to express fluorescent flashes during calcium spikes—which cause muscle to contract.
“We could see and measure in real time how blood vessels grew into the implanted muscle fibers, maturing toward equaling the strength of its native counterpart,” said Juhas.
The ultimate test will follow next, as the researchers need to investigate whether or not the biomimetic muscle can be used to repair actual muscle injuries and disease in humans.
“A number of researchers have ‘grown’ muscles in the laboratory and shown that they can behave in similar ways to that seen in the human body,” said Mark Lewis, an expert in skeletal muscle tissue engineering at Loughborough University, who wasn’t involved in the study. “However, transplantation of these grown muscles into a living creature, which continue to function as if they were native muscle has been taken to the next level by the current work.”
The findings were reported in the journal PNAS.
Was this helpful?



