Bacteria come in swarms, so managing their invasion requires a means of communication between individual bacteria. All sorts communication pathways are used to this end like chemical signals to make the bacterial swarm come to agreement and develop a pathogenic effect. By hijacking these pathways, it’s possible to avert infections and stop bacteria in its tracks, all without directly killing them with conventional means like antibiotics. Now, researchers in Germany report the discovery a previously unknown bacterial communication pathway, one that might be a potential target for new medicine.
Learning the ways of bacteria
Ralf Heermann, LMU
Dr. Helge B. Bode, an Merck-endowed professor for molecular biotechnology at the Goethe University in Frankfurt, and Dr. Ralf Heermann from the department of microbiology at the Ludwig Maximilian University in Munich previously discovered another bacterial communication pathway in 2013. In their most recent work, the German scientists a decoded a further new and widely distributed chemical type of bacterial communication.
Bacteria are known to communicate in nature primarily via the secretion and receipt of extracellular signalling molecules. This communication enables bacteria to execute sophisticated tasks such as dealing with antibiotic production and secretion of virulence factors.
In a way, bacteria can speak to each other, but instead of words as we know them the symbols used in bacterial communication consist of molecules. What is fascinating, is that scientists have not only decoded the syntax (the density of molecular symbols surrounding the bacteria), but have also begun to decode the meaning of the language used by bacteria, for example, in the case of a pathogen, communication that signals when to attack the host. This is just the beginning. We’re only scratching the surface.
[ALSO SEE] How bacteria control their size and remain uniform
The best known communication between bacteria identified so far happens via the N-acyl homoserine lactone (AHL). Through this pathway, an enzyme called Luxl produces and sends signals that are recognized by the LuxR receptor. When the chemicals bind to the receptor, bacteria get the OK to develop certain pathogenic properties and modulate their behavior towards one another. The process called “quorum sensing” needs a certain number of bacteria present to activate.
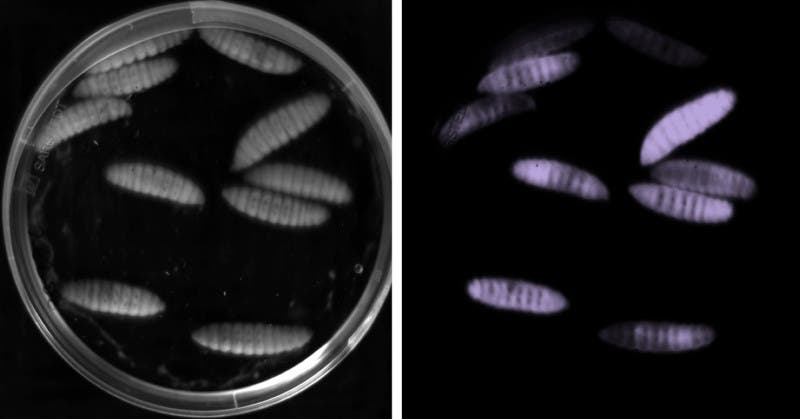
Credit: Ralf Heermann, LMU
The microbiologists looked at a strain of bacteria called Photorhabdus asymbiotica – a deadly pathogen for insects, which can also affect people causing skin infections. What’s odd about this strain is that it lacks a LuxR receptor, but not the enzyme Luxl. These bacteria communicate via the signal molecule dialkylresorcinol, which recognised the associated LuxR receptor.
“The influence on the pathogenic properties of the bacteria is at its strongest in this ‘quorum sensing’ system. P. asymbiotica requires dialkylresorcinol and in this way coordinates the communication with the conspecifics for the successful infection of the larvae”, says Helge Bode, whose group in 2013 also described the biosynthesis of this new signal molecule.
[MUST READ] Amazing bacteria that feeds on electricity lives on energy alone
Besides P. asymbiotica, the German microbiologists studied other bacterial species and found the pathway present there as well, signifying that it is widely distributed.
“We were able to identify several other bacteria that are pathogenic to humans that also do not express Luxl and also possess this ability for forming these signals”, says Heerman.
If the relevant communication options are prevented, the bacteria cannot develop their pathogenic properties. This means infections can be warded off by developing medicine and treatment that targets these pathways, all without the need to use antibiotics. The world is currently facing a challenging tolerance to antibiotics. Millions, even billions of people could become vulnerable to infections and antibiotic drugs would have no effect. People would then die from the most silliest diseases like they had only a hundred years ago.
“When pathogens are no longer destroyed by antibiotics as we have seen to date, but rather be impaired beforehand the formation of the pathogenic properties, the danger of resistance development would be substantially reduced”, says Bode.
Science reference:
Sophie Brameyer, Darko Kresovic, Helge B. Bode, Ralf Heermann.Dialkylresorcinols as bacterial signaling molecules. Proceedings of the National Academy of Sciences, 2015; 112 (2): 572 DOI: 10.1073/pnas.1417685112



