Renewable energy is clean, getting cheaper by the day and in many respects becoming more efficient thanks to rapid advancements coming from the world’s top-notch labs. It has one major drawback – storage. Before people can find a clever and cost-effective way to store all of that excess energy from wind and solar farms, chances have it that very few countries will want to pass the 30% renewables share margin. The best solution might actually be an old idea revamped to work for a novel setting – batteries. Not just any batteries though. The most promising solution so far is using flow batteries – storage mediums that are rechargeable and whose electroactive materials are stored external to the actual battery. Such a flow battery was developed at University of Southern California, and it might be a real game changer if the proper attention is awarded.
An elegant solution for a most troublesome problem
“The batteries last for about 5,000 recharge cycles, giving them an estimated 15-year lifespan,” said Sri Narayan, professor of chemistry at the USC Dornsife College of Letters, Arts and Sciences and corresponding author of an open-access paper published online by the Journal of the Electrochemical Society.
“Lithium ion batteries degrade after around 1,000 cycles, and cost 10 times more to manufacture,” he said.
These batteries aren’t meant to fit in your mobile, though. They’re intended for large-scale energy storage, backing power generating plants and making the grid more resilient. Where they shine, however, is in their potential to change how renewable energy is perceived in the broader picture. Solar panels can only generate power when the sun’s shining, and wind turbines can only generate power when the wind blows. Often, these systems are designed to produce just about as much energy as the grid can handle or just enough so it can power consumers, never more though. This makes renewables inherently unreliable. If you can find a way to cheaply and effectively store this excess energy, that a lot of people might consider their stance over renewable energy.

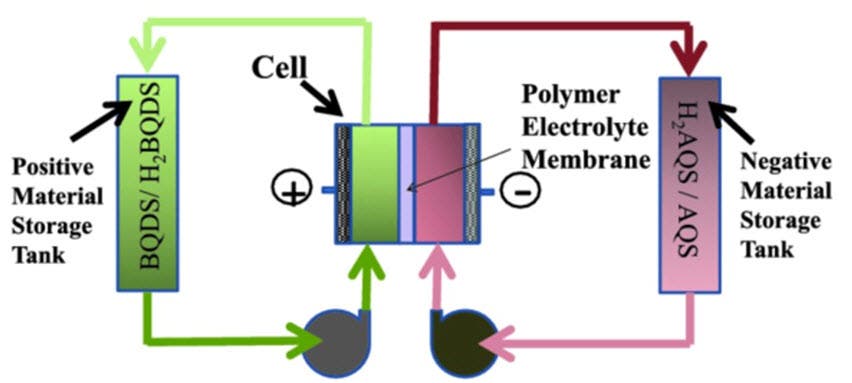
The working principle of the new battery, called the aqueous organic redox flow battery (ORBAT), is typical. It consists of a redox-flow design, very similar to a fuel cell, where two tanks of electroactive materials dissolved in water are pumped into the cell. Here, a proton carrying membrane separates the two fluids with electrodes on either side, releasing energy. The innovation lies in the tanks’ contents.
Such batteries typically work using metals, most often than not the toxic variety, as well as a precious metal catalyst. The ORBAT employs organic compounds dissolved in water and doesn’t employ an expensive catalyst. Using molecule design and a lot of trial-and-error, the researchers were able to find the perfect organic mix: quinones, or oxidized organic compounds. Quinones are found in plants, fungi, bacteria, and some animals, and are involved in photosynthesis and cellular respiration. Basically, Narayan and team modeled nature’s energy transfer model for their battery, and it works like a charm. Worth noting that similar solution was applied by Harvard researchers, but their prototype was only tested for about 100 cycles.
Currently, the quinones used by the ORBAT come from hydrocarbons, but there are ways to derive them directly from carbon dioxide. So there you have it: a scalable, inexpensive and sustainable metal-free rechargeable battery for large-scale electrical energy storage. But will we hear again about it four years from now? Fingers crossed.



