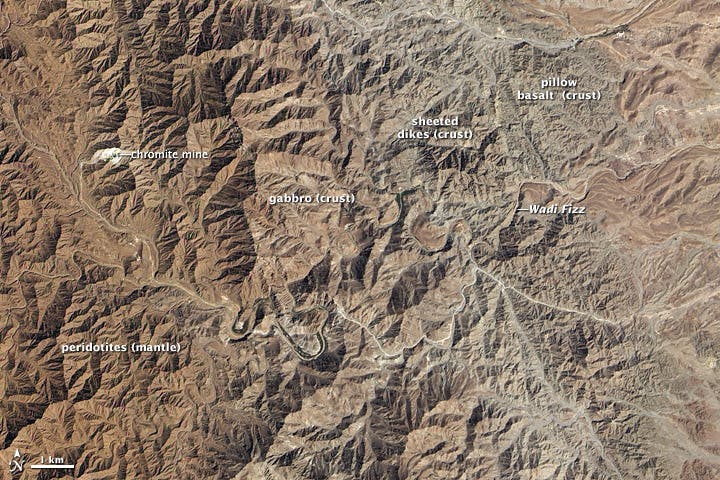
Deep inside the jagged red mountains of Oman, visitors with a trained eye might be shocked by an amazing sight: the world’s only exposed sections of the Earth’s mantle. Scientists are now sampling cores from this region to find out how a spontaneous process turned CO2 into limestone and marble millions of years ago. The answers they might unravel could one day offer a cheap and efficient solution for removing CO2 from the atmosphere and oceans.
Capturing carbon
The average American needs around 980 trees to offset their carbon footprint. Massive reforestation, especially in those hard struck parts of the world, must be a top priority in any policy meant to tackle climate change. But the truth is after you add the numbers, trees aren’t enough. We’re simply releasing more carbon from beneath the ground in the form of fossil fuels than any large forest can absorb. This is why many scientists are so interested in carbon capture and storage (CCS) technologies.
One of the most promising CCS projects I’ve come across is the Hellisheidi geothermal plant in Iceland. Here, carbon dioxide is dissolved in water and injected into the ground where it interacts with the surrounding rock and becomes mineralized. The whole process is 95 to 98 percent efficient and CO2 can basically turn into a rock in just two years.
Elsewhere, in China, the Sinopec fertilizer plant filters carbon and reuses it as fuel. According to the International Energy Agency, overall some 27 million tons of carbon are currently captured and stored across the world by 16 projects. An impressive start but in the grand scheme of things, that’s barely a dent (0.1%) in the 40 billion tons or so that human activity emits yearly.
“Any one technique is not guaranteed to succeed,” said Stuart Haszeldine, a geology professor at the University of Edinburgh who serves on a U.N. climate body studying how to reduce atmospheric carbon, for the Associated Press. “If we’re interested as a species, we’ve got to try a lot harder and do a lot more and a lot of different actions,” he said.
Naked Earth and its submerged mountains
One unique pathway for mitigating carbon in the atmosphere might be hiding in plain sights, in the rock formations of the al-Hajjar Mountains of Oman. The rugged mountains stand at their highest at 2,500 meters (8,200 feet) above sea level. Millions of years ago, some parts of these mountains were below the sea level. Many rocks used to lie in Earth’s interior, at the boundary between crust and mantle, but when the ancient Tethys Ocean narrowed and closed, the squeezing force thrust the ancient sea floor upward. This sort of landform is known to geologists as ophiolite, a term that describes oceanic crust that now sits at the land surface.
“You can walk down these beautiful canyons and basically descend 20 kilometers (12 miles) into the earth’s interior,” said in a statement Peter Kelemen, a geochemist at Columbia University’s Lamont-Doherty Earth Observatory, who has been exploring Oman’s hills for nearly three decades.
There are various rocks that comprise the ophiolite sequence. Along the boundary with the base of the crust, you can find chromite which the sultanate mines. Elsewhere, some layers of rocks (the first in the crust, rather than the mantle) are composed of gabbro. But what interests climate scientists is the peridotite, a rock that was once part of Earth’s mantle and reacts with the carbon in air and water to form marble and limestone.
It’s believed a billion tons of CO2 is trapped in the peridotites of al-Hajjar Mountains. When rain pulls carbon from the exposed mantle, stalactites and stalagmites form in the mountain caves.
In order to understand how all of this process works, Kelemen and 40 other scientists have formed the Oman Drilling Project. So far, $3.5 million in funding has been pledged by organizations around the world, among which NASA.
So far, the team has dug up dozens of core samples from four different sites in Oman’s mantle-exposed mountains. Some 13 tons of core samples will be shipped to the Chikyu, a research vessel off the coast of Japan where geologists will work round-the-clock shifts to better understand the chemical process that traps the carbon in rock. They’ll also be interested in finding ways to speed up the process given all that carbon has accumulated over 90 million years and time is not really on our side. In only 150 years, average CO2 levels in the atmosphere have soared from 280 to 405 parts per million.
“Just like in Oman’s mountains, the submerged rock would chemically absorb carbon from the water. The water could then be cycled back to the surface to absorb more carbon dioxide from the atmosphere. However, that would be following years of rigorous testing, but hopefully this project, and this discovery retains support,” Kelemen told the Oman Times.
Late 2015, 196 nations signed and later ratified the Paris Agreement — an international pact where each signatory pledges to cap or reduce its carbon emissions such that collectively we might steer away from any pathway that leads to more than 2 degrees C of warming. The elections in the United States have side tracked a bit this landmarked agreement, though officially all the other signatories besides the United States have vowed to stay steadfast and deliver on their pledges.
According to some estimates, however, the planet is already locked into 1.5 degrees C of warming. Renewable energy and energy efficiency measures, coupled with the scrapping of coal, oil, and gas, are paramount if we want to avoid catastrophic runaway global warming. But seeing how CO2 can stay in the atmosphere for up to two centuries, these measures have to be joined by CCS. With every passing day, CCS becomes more and more important. Hopefully, al-Hajjar’s secrets might help save us and the countless other species we share this planet with.