Magma forms much deeper than geologists previously believed, according to a new study conducted by Rice University.
Magma and Crust
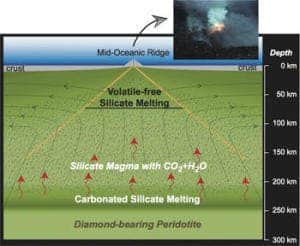
The group led by geologist Rajdeep Dasgupta put very small samples of peridotite under very large pressures, to find out if the rock can liquify, at least in small amounts, as deep as 250 km beneath the ocean floor. Peridotite is the dominant rock of the Earth’s mantle above a depth of about 400 km, and it was previously believed that the mineral doesn’t liquify above that depth.
The Rice team focused on mantle beneath oceans because that is where the crust is typically formed; in a much-simplified version, the silicate melts (magma) rise with the convective currents, cool as they reach the crust and then solidify to create new crust. The starting point for melting has long been thought to be at 70 kilometres beneath the seafloor.
Geophysics
In order to determine the mantle’s density and properties, geophysicists used seismic information; seismic waves from earthquakes travel the globe much like sound waves, and by measuring their speed, we can estimate some the medium’s properties. These waves travel faster in solids (especially in denser solids), and slower in liquids; the first questions arose here.
“Seismologists have observed anomalies in their velocity data as deep as 200 kilometers beneath the ocean floor,” Dasgupta said. “Based on our work, we show that trace amounts of magma are generated at this depth, which would potentially explain that.”
So it didn’t really add up – and this wasn’t the only clue. Geophysicists have also struggled to explain the bulk electrical conductivity of the oceanic mantle – something which was observed but couldn’t really be figured out.
“The magma at such depths has a high enough amount of dissolved carbon dioxide that its conductivity is very high,” Dasgupta said. “As a consequence, we can explain the conductivity of the mantle, which we knew was very high but always struggled to explain.”
The thing is, we cannot really dig down to the mantle – this is miles away from happening, both figuratively and literally, so we have to rely on indirect measurements (seismology, electric measurements, etc), lab experiments and surface extrapolations. Another interesting they found in this experiment was that carbonated rocks melt at significantly lower temperatures than non-carbonated.
“This deep melting makes the silicate differentiation of the planet much more efficient than previously thought,” Dasgupta said. “Not only that, this deep magma is the main agent to bring all the key ingredients for life — water and carbon — to the surface of the Earth.”
Volcanic windows
However, Dasgupta believes that volcanic rocks are the key to understanding our planet’s mantle.
“Our field of research is called experimental petrology,” he said. “We have all the necessary tools to simulate very high pressures (up to nearly 750,000 pounds per square inch for these experiments) and temperatures. We can subject small amounts of rock samples to these conditions and see what happens.”

To subject the rocks to these hellish conditions, they use massive hydraulic presses.
“When rocks come from deep in the mantle to shallower depths, they cross a certain boundary called the solidus, where rocks begin to undergo partial melting and produce magmas,” Dasgupta said. “Scientists knew the effect of a trace amount of carbon dioxide or water would be to lower this boundary, but our new estimation made it 150-180 kilometers deeper from the known depth of 70 kilometers,” he said.
These findings have major implications for all planetary sciences:
“What we are now saying is that with just a trace of carbon dioxide in the mantle, melting can begin as deep as around 200 kilometers. And when we incorporate the effect of trace water, the magma generation depth becomes at least 250 kilometers. This does not generate a large amount, but we show the extent of magma generation is larger than previously thought and, as a consequence, it has the capacity to affect geophysical and geochemical properties of the planet as a whole.”
The paper will be published this week in Nature
Was this helpful?