All biological processes on Earth rely on the sun for energy. It’s the sun’s rays that allow plants or cyanobacteria to grow. They become lunch for different creatures which in turn are preyed on higher up the food chain. Plants use the energy from incoming photons in a biological process known as photosynthesis, and for all intents and purposes, this is an elegant solution. However, it’s not all that efficient and researchers at the University of California, Berkeley, think they can come up with something better — or at least something different that might work well for some applications.
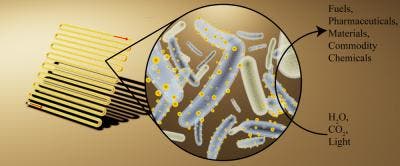
At the 254th National Meeting & Exposition of the American Chemical Society (ACS), Kelsey K. Sakimoto and colleagues showed off their latest attempt at harvesting energy with a hybrid system comprised of bacteria and what can only be described as tiny solar panels. Essentially, when the Moorella thermoacetica bacteria, which is nonphotosynthetic, was fed cadmium and the amino acid cysteine, it synthesized the food into cadmium sulfide (CdS) nanoparticles. These are the semiconducting materials many solar panels employ on their surface to collect photons and create electron-hole pairs.
They then showed that the hybrid system comprised of M. thermoacetica-CdS could make acetic acid from CO2, water, and light. “Once covered with these tiny solar panels, the bacteria can synthesize food, fuels, and plastics, all using solar energy,” Sakimoto said in a statement.
Plants turn CO2, water, and light into oxygen and sugars mainly through chlorophyll, which are the green pigments plants use to harvest sunlight. They’re quite similar to semiconductors employed in solar energy only photovoltaic cells turn all of that sunlight into flowing electrons whereas photosynthetic plant cells turn it into plant food. The problem is photosynthesis doesn’t seem all that efficient. Typically, most plants have a sunlight to biomass conversion of only 0.1%-0.2% whereas some crops see 1-2% efficiency.
The hybrid system, however, operates at an efficiency of more than 80 percent, all in a self-replicating and self-generating environment. “These bacteria outperform natural photosynthesis,” Sakimoto said.

Of course, none of this makes trees and plants obsolete. Given our urgent need to phase off fossil fuels, however, any alternative technology that can generate clean energy or products is more than welcome. Acetic acid, for instance, is a very versatile chemical. It’s widely used in the chemical manufacturing industry to make polymers, pharmaceuticals, even liquid fuels. In fact, you have a 5-20% acetic acid-water solution in your kitchen right now — vinegar. Even among alternative energy systems, such as artificial photosynthesis devices, this bacterial-semiconductor system offers long-standing benefits.
“Many current systems in artificial photosynthesis require solid electrodes, which is a huge cost. Our algal biofuels are much more attractive, as the whole CO2-to-chemical apparatus is self-contained and only requires a big vat out in the sun,” Sakimoto points out.
For now, he and colleagues are working on making the semiconductor and bacteria interact better. They’re also looking at other matches that might render different chemicals or foods.
Was this helpful?