Known as tafenoquine, the new drug was developed by GlaxoSmithKline (GSK). After receiving FDA approval, the drug also has to pass the scrutiny of other national drug regulation agencies before it can see global use.
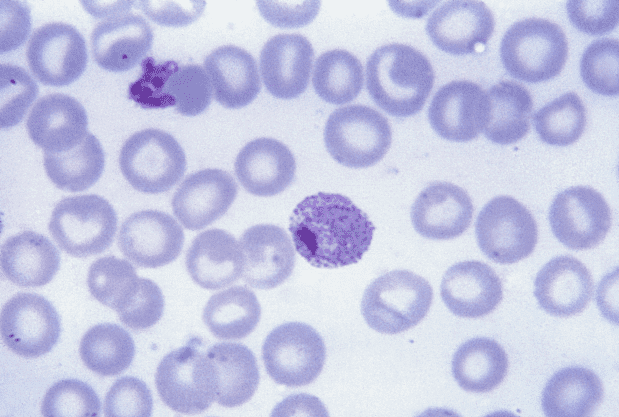
Image credits CDC / Steven Glenn, Laboratory & Consultation Division.
The compound is tailored to fight the recurring form of malaria. This disease, caused by the parasite Plasmodium vivax and estimated to infect over 8 million people each year, is especially tricky to cure as the parasite can hide in the host’s liver and emerge many times over a period of years.
Persistent malaria
The recurring form of the disease is the most commonly encountered type of malaria outside of Sub-Saharan Africa. Children are particularly hard-hit by the disease. A single bite from a malaria-infected mosquito will cause several bouts of the disease, which means a lot of missed school and progressively-poorer health during formative years.
Even worse, infected individuals act as unwilling spreaders of the disease. If the parasite reawakens and passes through to their blood, any mosquito that bites them will spread malaria to other people. This combination of a dormant state and high infectivity make recurring malaria especially hard to eliminate.
Still, with the FDA’s recent approval of tafenoquine, that state of affairs may change. The drug can flush the parasite out of its dormant state in the liver, preventing it from re-infecting its host. Even better, tafenoquine doesn’t interfere with other drugs used to treat the immediate infection caused by the parasite, so both treatments can be taken side-by-side.
We already have a drug on hand to clear the plasmodium out of the liver — it’s called primaquine. However, primaquine treatment needs to be followed for 14 days at a time, while the tafenoquine course only involves taking a single dose.
There’s a legitimate concern in the medical community that many patients with recurring malaria feel better after a few days of primaquine treatment and don’t follow the course to its completion — which isn’t nearly enough to flush out all the parasites.
While the FDA approval of the drug vouches for its effectiveness, the administration warns that the drug comes with side effects you should be aware of.
First and foremost, people suffering from enzyme G6PD deficiency should not take the drug, as it can cause severe anemia. The FDA recommends that patients are screened for this deficiency before being given the treatment — but this may be virtually impossible in the poorer areas where malaria is most common. Secondly, there are concerns that higher doses may interact with pre-existing psychiatric conditions.
Despite its potential risks, the drug could help reduce the incidence of Plasmodium vivax malaria on a global level, however.
“The approval of Krintafel [the brand name for tafenoquine], the first new treatment for Plasmodium vivax malaria in over 60 years, is a significant milestone for people living with this type of relapsing malaria,” says Dr Hal Barron, president of research and development at GSK. “Together with our partner, Medicines for Malaria Venture, we believe Krintafel will be an important medicine for patients with malaria and contribute to the ongoing effort to eradicate this disease.”
The next step will be for the drug to be assessed by regulators in countries where this form of malaria is a significant problem.
Was this helpful?



