It’s hard to image a world without colours simply because they’re all around us. Have you ever wondered, though, where do colours come from? To answer this question, we first have to understand how human colour perception works and how matter physically interacts with light.
What gives colour

White light is a mixture of all colours, including those that the human eye can’t see. When we say something has colour, what we actually mean is that light of a particular range of wavelengths is reflected more strongly than the light of other wavelengths. How matter behaves in the presence of light, consequently appearing coloured to us humans, depends on a couple of major factors. First of all — everything is made up of electrons and atoms, but each substance has a different number of atoms and different electron configuration. This way, when light hits matter one or more of the following phenomena happens:
- reflection and scattering. Most objects reflect light, but some are more reflective than others, like metals. This is directly related to the number of free electrons that are able to pass from atom to atom with ease. Instead of absorbing energy from the light, the free electrons vibrate and the light energy is sent out of the material at the same frequency as the original light coming in.
- absorption. When there’s no reflection (the object is opaque), then the incoming light source frequency is the same as, or very close to, the vibration frequency of the electrons in the given material. The electrons thus absorb most of the incoming energy, with little or no reflection.
- transmission. If the incoming light energy is much lower or much higher than that required for the electrons comprising an object to vibrate, then the light source will pass through the material unchanged. This way matter will look transparent to the human eye, such as in the case of glass.
- refraction. If the energy of the incoming light is the same as the vibration frequency of the electrons in the material, light is able to go deep into the material, and causes small vibrations in the electrons. The vibrations are then passed on from atom to atom, each vibrating at the same frequency as the incoming light source. This makes the light inside the material look bent. Example: a straw in a glass of water.
Light and matter
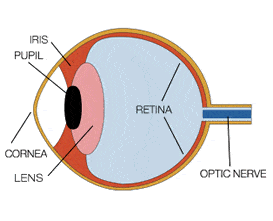

Image via Pantone.com
The human eye and brain translate light into colour. Light receptors within the eye transmit messages to the brain, producing the familiar sensation of colour. The retina is covered by millions of light-sensitive cells, some shaped like rods and some like cones, and it’s these receptors that process the light and then send this information to the visual cortex. Rods are mostly concentrated around the edge of the retina and transmit mostly black and white information. Cones transmit the higher levels of light intensity that create the sensation of color and visual sharpness. These cells, working in combination with connecting nerve cells, give the brain enough information to interpret and name colours.
Think of atoms like bricks in a wall (chemical compound). Imagine throwing a ball into the wall. If the wall is smooth or has sharp corners, the ball may jump back in different directions. However, if the wall is filled with holes, the ball may go through the wall or get stuck in one of the tricky corners, respectively. Same with every surface when light hits it. The surface may reflect the light back; it can absorb light or just let it pass through (transparent things).
This analogy is far from perfect though because light isn’t like a ball. For instance, the light we get to see, called visible light, is only a fraction of the full range of frequencies. A molecule might absorb photons from anywhere across the whole electromagnetic spectrum, from radio waves to X-rays, but it will be colourful only if there is a difference in how strongly it absorbs one visible wavelength over another. As it turns out, this is quite uncommon since most molecules absorb light above the visible spectrum, in the ultraviolet range. So, because electrons in most molecules are bound very tightly, most compounds are white!
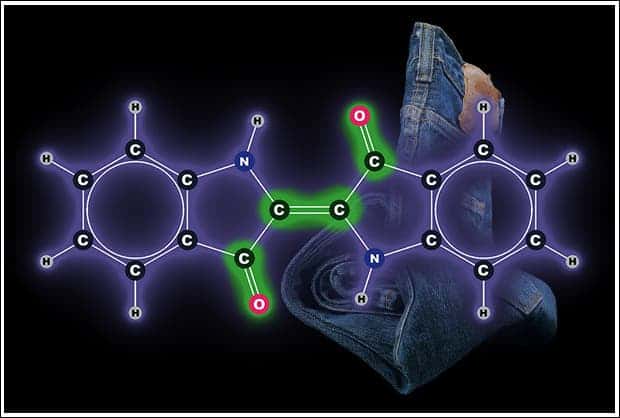
Some substances have electrons in the right range of binding strength which makes them suitable to use as dyes. One of the first natural dyes is indigo, commonly used to colour jeans. It derives its colour from a set of three double-bonds at its centre (O=C, C=C, C=O). The problem with indigo and other organic dyes is that it fades away in time because it absorbs energy, instead of reflecting it. In time, bonds break as a result of the damage. Inorganic dyes like pure iron oxide or rust (ochre), however, are lightfast and can last for thousands of years. This is why cave paintings are still visible today!

Lycopene absorbs most of the visible light spectrum and reflects mainly red back to the viewer, thus a ripe tomato appears red. Image: Colour Therapy Healing
As a conclusion, things do not have color by themselves — only when light (energy) hits them, we can see colors. This is precisely why your surroundings appear greyish or downright black when you’re in the dark. Also, remember our eyes can only see a limited range of colours. But dogs, cats, mice, rats and rabbits have very poor colour vision. In fact, they see mostly greys and some blues and yellows, while bees and butterflies can see colors that we can’t see. Their range of color vision extends into the ultraviolet, and in fact, they couldn’t have survived otherwise. Evolution led bees to adapt ultraviolet vision because flowers leave scatter ultraviolet patterns, allowing the insects to easily identify targets and pollinate. But while humans can’t see colours beyond our visible spectrum, the machines we build can. This is what spectrometers are for.






