Every bone in your body no older than ten years despite how old you might actually be. Just like skin, bone sheds old tissue and grows a new one from stem cells sourced from the bone marrow. Some scientists say that observing how these stem cells interact when they transform into bone tissue could lead to new treatment and drugs for diseases such as osteoporosis. Imaging stem cells inside bones, however, has always proven challenging — until now. Remarkably, American researchers recently showed how to grow intact and transparent bones.
You don’t feel like your bones are changing because the process involves a delicate interplay of cells that build new bone mass and cells that break down old bone mass. This continual remodeling cycle is controlled by stem cells called osteoprogenitors that develop into osteoblasts or osteocytes. These cells are what regulate and maintain the skeleton. This is why scientists find it crucial to understand how these stem cells behave when new bone mass is created. But this isn’t easy. Bones don’t lie but they sure don’t give up secrets easily. The stem cells are pretty rare and not evenly distributed throughout the bone.
To determine stem cell populations in the bone, researchers usually slice the bone into thin sections then extrapolate the number of stem cells. Not only does this method introduce a lot of uncertainties, the slicing also deteriorates the bone. Being able to peer inside the bone somehow is thus very desirable in the field.
A while ago, Viviana Gradinaru, assistant professor of biology and biological engineering at CalTech, helped develop a technique called CLARITY which can render soft tissue like the brain transparent. It manages this through the removal of lipids from cells which cause tissue to be opaque. Additionally, a clear hydrogel mesh is infused to provide structural support. The approach is so effective that at one point, while working as a post-doc at Stanford University, Gradinaru and colleagues were able to make all of the soft tissue inside a mouse transparent.
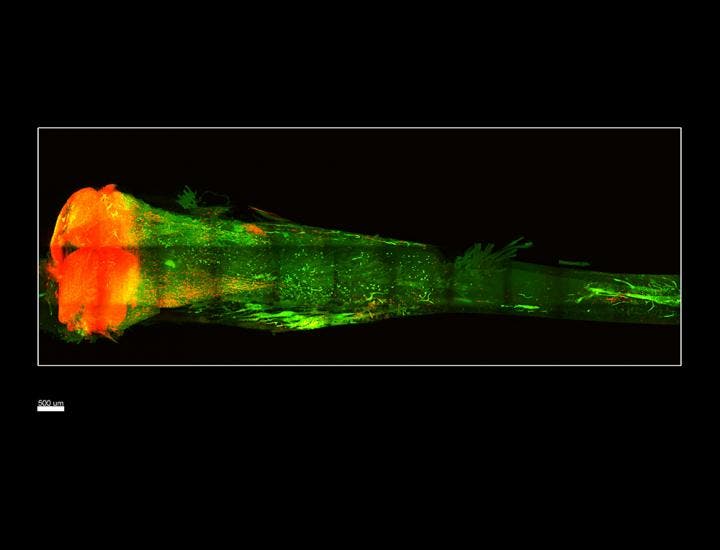
Now at CalTech, Gradinaru’s lab expanded the method so it works for hard tissue as well. The team first started with bones from postmortem transgenic mice which were genetically engineered to have red fluorescent stem cells so these could be more easily identified. The types of bones selected for the study were the femur and tibia, as well as some bones of the vertebral column. No bone was longer than a few centimeters, that’s for sure.
After they removed the calcium molecules from the bones which contribute to opacity, the team infused the bones with a hydrogel that locks cellular components in place and preserve the architecture of the sample. The last step involved using a detergent to flush away the lipids, leaving a transparent bone instead, as reported in Science Translational Medicine.
To image the cells inside the bones properly, a custom light-sheet microscope for fast and high-resolution visualization was employed. This instrument does not damage the fluorescent signal.
Already, the Bone CLARITY method is being used in collaboration with a biotech company to test a new drug for osteoporosis. The condition occurs when loss of bone mass leads to a high risk of fractures and affects millions of Americans yearly.
“Biologists are beginning to discover that bones are not just structural supports,” says Gradinaru, who also serves as the director of the Center for Molecular and Cellular Neuroscience at the Tianqiao and Chrissy Chen Institute for Neuroscience at Caltech. “For example, hormones from bone send the brain signals to regulate appetite, and studying the interface between the skull and the brain is a vital part of neuroscience. It is our hope that Bone CLARITY will help break new ground in understanding the inner workings of these important organs.”
Was this helpful?



