Only antimatter is more expensive than endohedral fullerenes which an Oxford startup just recently sold for $167 million per gram. According to Designer Carbon Materials – the only company in the world that manufactures this exotic material – it sold 200 micrograms of pure endohedral fullerenes for $33,400.

Endohedral fullerenes is basically a nitrogen atom encased by 60 carbon atoms. These carbon cages are called buckminsterfullerene and resemble the vertices on a football, which is why they’re also known as buckyballs.
Buckyballs could not exist without carbon’s versatility. Carbon is the most versatile element, allowing carbon molecules to take on many forms, or allotropes.
Molecules consist of multiple atoms chemically bonded together. These atoms can be different elements, as with water (H2O), but they do not have to be, as with oxygen (O2). Though, molecules with only one element are not restricted to just one shape. For example, oxygen can also form ozone (O3), which has a different shape than O2. Oxygen, in its O2 shape, is said to be a different “allotrope” to oxygen as ozone, in its O3 shape. Oxygen also has two other allotropes – O4 and O8 – that are a lot stranger, but far less common.
Meanwhile, carbon has over a dozen different allotropes. Unlike oxygen, these shapes do not just differ by the number of molecules in the allotrope. Instead, carbon allotropes are defined by fundamentally different structures. Here are some examples:
- Diamond – the most loved carbon allotrope – exists in a lattice shape with carbon molecules bonded at tetrahedral angles.
- Graphite – commonly found in pencil lead – exists as sheets or layers of hexagon tessellations stacked and bonded together.
- Graphene – which ZME Science frequently writes about for its many groundbreaking properties – exists as just one single sheet of graphite. Graphene also plays a role as an intermediate step in forming buckyballs in space.
- Fullerenes exist as a variety of “3-D shapes.” Fullerenes with 60 carbon molecules are one form of C60. These 60 molecules can be arranged as 20 hexagons interspersed with 12 pentagons. Can you picture that? This is exactly the same shape as a normal soccer ball, except that fullerenes are hollow and not manufactured by Adidas.
To make a molecule with 60 carbon atoms, you need to start with at least 60 carbon atoms. There are two possible ways to get 60 of these atoms. (1) As one option, you can start with a bunch of smaller molecules that each have a few carbon atoms and then combine them together. (2) Or as an alternative, you can start with a larger molecule with at least 60 carbon atoms and then break it apart.
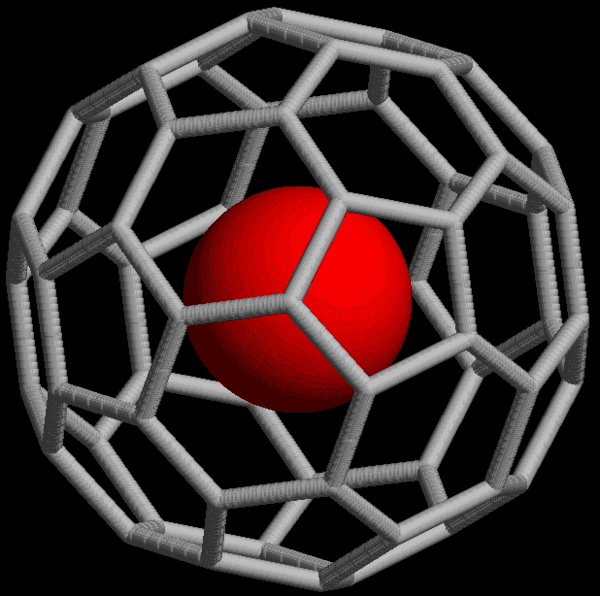
I have no idea how Designer Carbon Materials manufactured its endohedral fullerenes, but apparently it’s very difficult to make. It’s the only company in the world that makes N@C60. According to Ars Technica, the Oxford-based startup can make half a gram of the fullerenes in a day, but of much lower quality and purity. The premium N@C60 can be manufactured at 50 milligrams per day and takes weeks to purify. “We’re a new company, only starting to sell the product,” said Dr. Kyriakos Porfyrakis, the company’s founder. “But after the first sale we have more requests from around the world, so the demand seems to be increasing.”
Fullerenes molecules have extraordinary electrical properties. What makes the nitrogen variety so special (and ludicrous expensive), however, is a long electron spin lifetime. This makes them perfect for atomic clocks – an extremely accurate type of clock which is regulated by the vibrations of an atomic or molecular system. The auto industry is particularly interested in atomic clocks since these can be used to jump GPS accuracy to the millimeter range, which is essential if selfdriving cars are ever to become widespread and, most importantly, safe.
“At the moment, atomic clocks are room-sized,” said Lucius Cary, a director of the Oxford Technology SEIS fund, which now holds a minority stake. “This endohedral fullerene would make it work on a chip that could go into your mobile phone.
“There will be lots of applications for this technology,” he continued. “The most obvious is in controlling autonomous vehicles. If two cars are coming towards each other on a country lane, knowing where they are to within 2m is not enough but to 1mm it is enough.”
“Every mobile phone could one day contain one of these things,” Mr Cary claimed.




