In theory, all natural sciences can be reduced to physical forces. In practice, however, imaging and studying these physical forces becomes extremely difficult, especially in the case of small objects like cells. Now, for the first time, researchers have developed a tool to monitor the physical forces inside live cells.
Scientists from the University of Geneva (UNIGE) and the National Centre of Competence in Research (NCCR) in Chemical Biology, Switzerland, have developed probes capable of entering into cells and image physical forces.
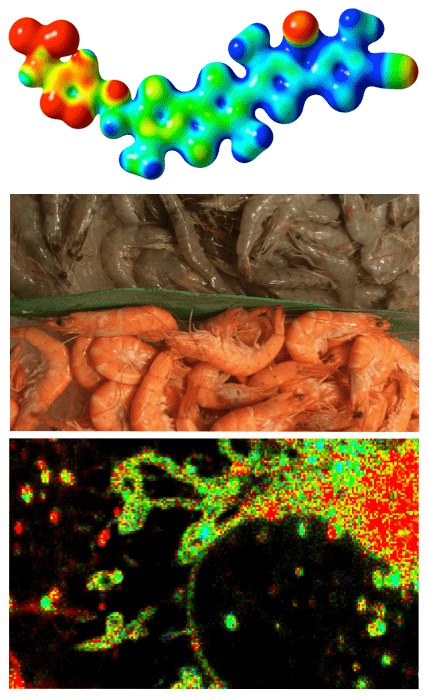
The research team took inspiration from an unlikely place: shrimp cooking. In live shrimp, the physical forces surrounding the protein flatten a pigment called astaxanthin. Astaxanthin is initially dark orange, but the flattening polarizes it until it turns blue. Cooking the shrimp makes the pigment return to its initial color. For 8 years, researchers have been trying to operate on the same principle of polarization — and now they’ve finally done it.
“Our approach to creating tension probes was inspired by the color change of shrimp, crabs or lobsters during cooking,” says Stefan Matile, Professor in the Department of Organic Chemistry at the Faculty of Science of the UNIGE and member of the NCCR. “During cooking, these proteins are unfolded and the lobster pigment can regain its natural dark orange color,” continues the Geneva chemist.
Last year, the team produced the first fluorescent probe capable of imaging the forces acting on the outer membrane (called the plasma membrane) of living cells. The study was so popular that requests from more than 50 laboratories around the world came immediately after the results were released.
However, there’s a big difference between analyzing the outside of cells and the inside of the cells. The outside can be accessed by a variety of tools such as micropipettes, optical clamps, etc. — whereas those tools cannot access the forces within cells, says Aurélien Roux, a professor in the Department of Biochemistry at the Faculty of Science of the UNIGE and a member of the NCCR.
“Organelles such as mitochondria, responsible for energy production; endoplasmic reticulum, responsible for protein synthesis; endosomes, responsible for trafficking material to and within cells; or the nucleus, which stores genetic information, are simply beyond the reach of physical tools from outside.”
This is where the new fluorescent probes come in, as a way to visualize all the forces that work within these organelles — offering us an unprecedented view of the cells’ inside.
“These new probes now offer us the opportunity to tackle mechanobiology and revolutionize the study of life sciences,” concludes Matile.
The study was published in the Journal of American Chemical Society (DOI: 10.1021/jacs.8b13189)



