The EpiPen is going generic.
Image credits Intropin / Wikimedia.
The U.S. Food and Drug Administration (FDA) approved the first generic version of the EpiPen and EpiPen Jr auto-injectors for allergic reactions. The devices, used to inject epinephrine in the emergency treatment of allergic reactions in adults, were mired in controversy as its producer, Mylan, applied ludicrous price increases to the life-saving devices.
Mylan’s CEO, Heather Bresch, eventually had to testify in front of the House Oversight and Government Reform Committee about the drug’s price increase back in 2016. You can read about the fallout in all its glory here and here.
The generic version should give patients an affordable alternative and break Mylan’s monopoly over this device. US-based Teva Pharmaceuticals gained approval to market its generic epinephrine auto-injector in 0.3 mg and 0.15 mg strengths. The company hasn’t provided any details about when the generics will hit the market or their planned price.
Off-brand, on point
“Today’s approval of the first generic version of the most-widely prescribed epinephrine auto-injector in the U.S. is part of our longstanding commitment to advance access to lower cost, safe and effective generic alternatives once patents and other exclusivities no longer prevent approval,” said FDA Commissioner Scott Gottlieb, M.D.
Patients with severe allergies can’t afford the luxury of not buying EpiPens; their life quite literally depends on it in the case of a severe allergic reaction (called ‘anaphylactic reactions‘). Life-threatening allergies can include reactions to insect bites or stings, foods, medications, latex, or other causes.
Allergic reactions typically affect a localized area of the body. Our immune systems react to a particular allergen, and specialized types of T and B (white) cells flood tissues with cytokine, interleukin-4, and antibodies in response; this is what causes allergic symptoms such as inflammation. Anaphylactic reactions take place pretty much the same way but affect the whole body. If left untreated, such a reaction can and often does lead to death.
Epinephrine (or adrenaline, it’s the same substance) works by reducing swelling in the airway and increasing blood flow in the veins.
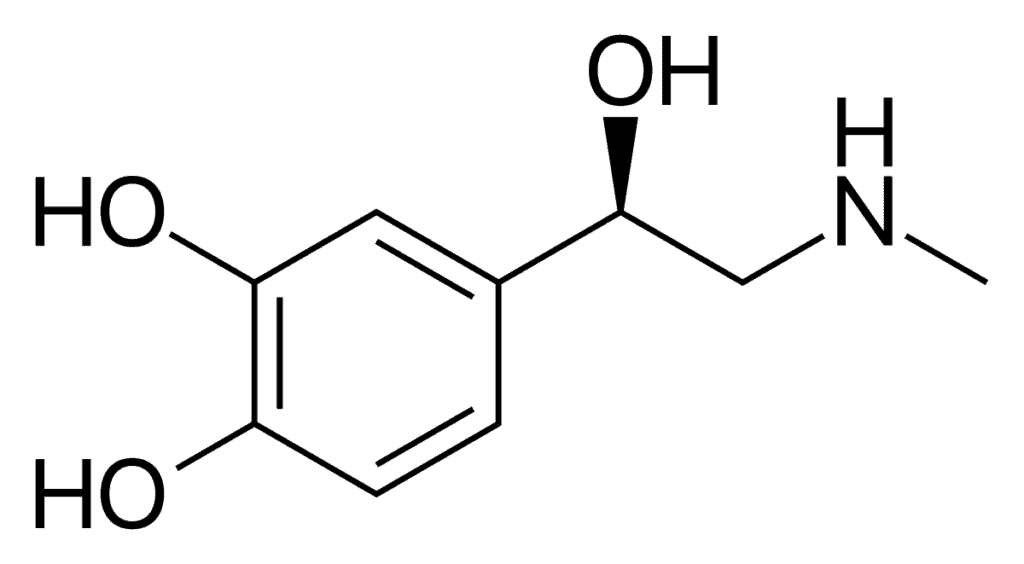
Image via Wikimedia.
Needless to say, this puts patients in a tight spot when price hikes leave them — again, quite literally — unable to afford the devices. The FDA’s move is meant to protect exactly these people. Patients with severe allergies now have a lower-cost option for the treatment they need. Approval for the generic version should also “help protect against potential drug shortages”, the FDA explains.
“We remain committed to doing our part to provide scientific and regulatory clarity for sponsors seeking to develop complex generics, as well as prioritize the approval of medicines with little or no generic competition as part of our overarching effort to remove barriers to generic development and market entry of critically important medicines,” the Administration adds.
Products such as the EpiPen are known as ‘combination products’ — they consist of both a drug (epinephrine) and a device (the autoinjector or ‘pen’ itself). They’re more complicated to develop compared to regular drugs, and the FDA regularly takes steps to help guide industry through the process. The FDA dutifully requires adequate proof that combination products produce consistent, quality results before giving them the seal of approval.
However, this (alongside patent rights) also means that it’s hard for other agents to come up with a competitor product for even relatively simple devices such as the EpiPen. To their credit, the FDA is making efforts to streamline the process of getting a generic approved — for example, they announced the Drug Competition Action Plan last year, outlining the steps required to get such a generic to market.
And it seems to be working. The generics are the first competitors cleared by the FDA that are direct generic copies of the EpiPen. “Authorized generics” are basically the same as the branded product but don’t display the brand or manufacturer on their package. Companies may choose to sell an authorized generic at a lower cost than the brand-name drug product.
Other epinephrine auto-injectors, such as the Adrenaclick and Auvi-Q, are on the market but aren’t considered EpiPen generics.
“We’re applying our full resources to this important launch in the coming months and eager to being supplying the market,” Teva said in a statement.
Roughly one in 50 Americans experience anaphylaxis, according to the Anaphylaxis and Allergy Foundation of America. It’s not a one-time affair, either; anyone who goes through one anaphylactic episode always faces the risk of going through another one. These patients thus always carry an emergency dose of epinephrine with them at all times; many keep more than one dose handy.
The EpiPen conquered the market because it’s simple and quick to administer. All you have to do is take the pen, jam it in your thigh, and it automatically injects a dose of epinephrine to stop the allergic reaction.
Was this helpful?



