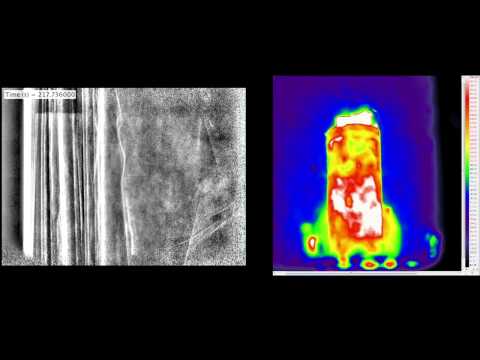
Lithium-ion batteries have pervaded most mobile technologies, including phones, notebooks or electric vehicles. Scientists involved in lithium-ion batteries are mainly interested in increasing the energy density so they can last longer and accelerating the charging time, but also avoiding failures. You can watch on YouTube a myriad of such fails, like batteries exploding and such. Thankfully, these events are particularly rare, yet they signal there’s still much room for improvement. University College London researchers were interested in studying how lithium-ion batteries perform under a certain kind of stress resulting from overheating, and recorded the first thermal failure using thermal imaging and non-invasive high speed imaging techniques to observe the internal structure. This way, they recorded both what happens outside and inside the battery when it overheats.

This phenomenon is in fact called thermal runaway in literature – basically, it occurs when more heat is coming into the system than it exists. Paul Shearing and colleagues took two commercially available lithium-ion batteries, dubbed Cell 1 and Cell 2, then subjected them to an external heat source. After 2 minutes the Cell 1 started venting molten material into its surrounding environment. Copper material inside Cell 1 melted, indicating internal temperatures of at least 1,085°C. As for Cell 2, the rapid pressure rise (temperature is directly proportional to pressure) caused its top lid to blow off entirely. In real-world applications, this additional oxygen coming through the open lid means even more thermal runaway. The videos below show how the two cells fail under the thermal load.

CELL 1

CELL 2
[RELATED] New lithium ion battery cathod can withstand 25,000 cycles. Your laptop battery only has 300
The authors found that thermal and electrochemical reactions inside both cells produced gas pockets that deformed the spiral wound layers of the cells. Cell 2 was the most compromised of the two batteries, structurally. Unlike Cell 2, Cell 1 was designed with a central cylindrical support, which seemed to help maintain structural integrity. The findings appeared in Nature; hopefully these will help design better lithium-ion batteries. Explosions are scary! Let’s have less of those…
Was this helpful?