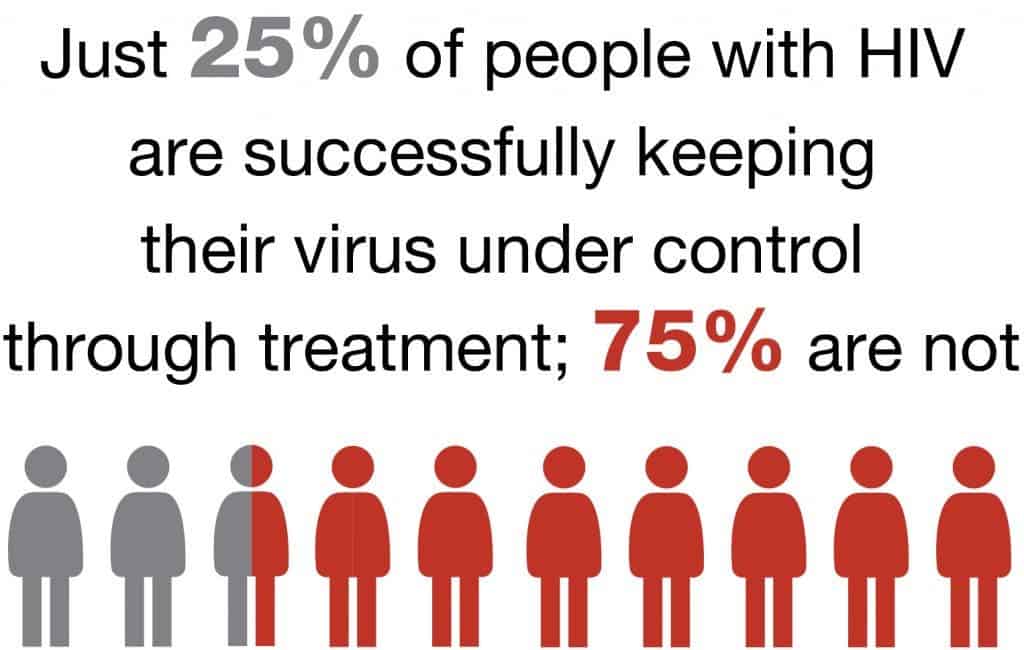
Two new studies – both covering gay men, one in Britain and the other in France – were recently shared with the public boasting terrific results. In the trials, gay men were asked to take a drug called Truvada either daily or right before and after having sex. In an unlikely event of chance, both studies found a 86 percent reduction in new HIV infections among volunteers using Truvada. This suggests that the orally administered drug might be a lifeline in many HIV-ridden communities, considering 90% of all HIV cases could be prevented if those infected seek treatment.
ZME science first reported about the potential benefits of Truvada a few years back. It has since received FDA approval.
The first trial, called IPERGAY, included 400 men who engaged in high-risk sex with other men in Canada and France. Volunteers were divided into two groups: one would receive Truvada pills, the other placebos. The men took two pills between two and 24 hours before sex, and a third and fourth pill 24 and 48 hours later. On average, the men involved had sex four times a week. There were two new HIV infections among men taking Truvada, and 14 new infections among those taking the placebo pills, which means an 86% reduction of the risk of contacting HIV.
The second trial, called PROUD, involved more men – 545 gay and bisexual men in the United Kingdom, all of whom having admitted they’had unprotected sex recently. Half were assigned to begin taking Truvada every day right away, while half were assigned to receive Truvada after one year (almost like a placebo). In the group that started Truvada right away, three men contracted HIV. However, in the group that delayed Truvada, there were 19 new HIV infections. That’s a risk reduction of 86 percent for the first group, just like the IPERGAY trial found.
“These results are extremely exciting and show PrEP [pre-exposure prophylaxis] is highly effective at preventing HIV infection in the real world,” said Sheena McCormack, a professor of clinical epidemiology at University College London and chief investigator of the PROUD study. “These results show there is a need for PrEP, and offer hope of reversing the epidemic among men who have sex with men.”
Unfortunately, the results for Truvada won’t necessarily work for heterosexual couples, since HIV is transmitted differently based on the kind of sex partners have. But if it can work for gay couples, maybe some other drug will for heterosexuals as well. Either way, the results are extremely heartening. In fact, the drug worked so well that the researchers had to cut the program off because it was unethical to deprive the control group of a preventive treatment that obviously works. Both studies were presented at the at the Conference on Retroviruses and Opportunistic Infections in Seattle, Washington.
Less encouraging is another study which found that a $2.8 million three-year long program which paid some 9,000 patients in 39 clinics to get treated rendered little to no results. Clinics where money was distributed did only 5 percent better than those that did not, which is marginally insignificant statistically-wise. The government paid HIV patients in Bronx and in Washington — where infection rates are high among poor blacks and Hispanics — up top $280 a year to take pills which would not only help them with their symptoms, but also slow down infection to other healthy people.
“We did not see a significant effect of financial incentives,” said Dr. Wafaa M. El-Sadr for the New York Times, an AIDS expert at Columbia University and the lead investigator. But, she said, there is “promise for using such incentives in a targeted manner.”
Was this helpful?



