A non-invasive hand-held device could make life a lot more bearable for patients suffering from idiopathic gastroparesis. The device delivers transcutaneous vagal nerve stimulation, relieving symptoms and accelerating gastric emptying.
Gastroparesis is a medical condition that affects the normal spontaneous movement of the muscles in your stomach. In normal conditions, the stomach’s muscle contractions move food through the digestive tract. However, patients with gastroparesis have a much weaker stomach motility, preventing the stomach from emptying properly.
The most common symptoms of gastroparesis include:
- Bloating;
- Nausea;
- Stomach pain;
- Vomiting;
- Acid reflux;
- Feeling full after just a few bites;
- Weight loss due to malnutrition;
- Changes in blood sugar levels;
Gastroparesis can cause a lot of health complications. Besides annoying cramps and abdominal pain, the condition causes many patients to become malnourished since even a few bites can make them feel full. Food that remains for too long in the stomach can also harden into a solid mass, known as a bezoar. In some cases, this can be fatal when blocking the small intestines.
There are factors that can increase the risk of having Gastroparesis, including:
- Being infected by a virus;
- Low thyroid (when your glands do not produce enough of a certain hormone);
- Having a connective tissue disease;
- Diabetes;
- A nervous system disease, like Parkinson’s and others;
- Medication, like those that slow down the rate of stomach emptying.
Usually, doctors advise an immediate change in diet in order to start treating the condition. One first step in managing gastroparesis involves eating smaller meals more frequently and chewing food thoroughly. Patients will also be prescribed medication that stimulates the muscles of the stomach and controls the reflux. In extreme cases, surgery may be necessary in order to install a gastric venting tube, which relieves pressure in the gastric tract.
Now, patients suffering from gastroparesis have a new avenue of treatment at their disposal that is non-invasive and easy to implement.
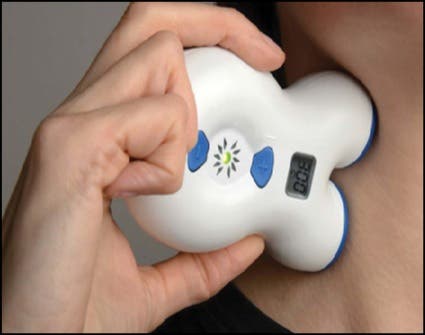
The procedure, known as vagal nerve stimulation (nVNS), involves a gammaCore hand-held vagal nerve stimulator (electroCore). The device is already approved by the FDA and was originally designed for treating migraines and cluster headaches.
The effectiveness of the device for treating gastroparesis was recently assessed in a pilot study presented at the 2019 annual meeting of the North American Neuromodulation Society.
“Transcutaneous VNS is a promising noninvasive technique with a potential to improve both subjective and objective clinical outcomes in patients with gastroparesis,” said Ronnie Fass, MD, the director of the Esophageal and Swallowing Center at MetroHealth Medical Center in Cleveland, who was not involved in the research. “The device was able to improve an array of gastroparesis-related symptoms, the Gastroparesis Cardinal Symptom Index [GCSI] and gastric emptying. While these results are very exciting with a clear potential for making this device part of our therapeutic armamentarium for gastroparesis, a larger study is needed to establish the value of this device in clinical practice.”
Previously, a study published in Frontline Gastroenterology found that out of 23 gastroparesis patients who tried nVNS, eight experienced significant symptom improvement (35% success rate). In the new pilot study, a team led by Dr. Linda Nguyen, of the Division of Gastroenterology and Hepatology at Stanford University School of Medicine, assessed the effectiveness of nVNS for gastroparesis symptoms as well as gastric emptying and autonomic function.
The study involved 15 patients with idiopathic gastroparesis. All participants self-administered nVNS twice a day for four weeks, followed by a four-week washout period. Each daily treatment involved using the device for two minutes on each side of the neck. With each treatment, the voltage was increased until the lip twitched to indicate that the facial nerve was engaged.
Six patients (40%) responded well to the treatment, showing significant improvements in all cardinal symptoms of gastroparesis. These improvements persisted after the four-week washout period but were not as significant as immediately following the treatment. The VNS therapy also accelerated gastric emptying by 26 minutes (half time), the researchers reported.
These are very promising results, although some critics have highlighted some of the study’s limitations. For one, this wasn’t a controlled trial, so the participants may have experienced a powerful placebo effect. The clinical effects aren’t very robust either, meaning the effect size is rather small. In the future, nVNS might become a useful complementary treatment but it won’t likely be considered a stand-alone treatment option. Lastly, the study shows that not everyone can benefit from nVNS.



