A major breakthrough in the production of lab-grown meat promises to make the technique even more affordable and attractive. Researchers in Singapore have used magnetic fields to replace the need for fetal bovine serum (FBS) from dead cows. This approach, they say, offers a novel way of growing cell-based meat.

When the first lab-grown burger was produced, it cost over $250,000. Less than ten years later, the first restaurant selling lab-grown meat was opened in Singapore and several countries already have factories on standby, just waiting to start.
Lab-grown meat is appealing for several reasons. For starters, it’s ethical — you don’t need to kill animals (or rather, you need to kill way fewer animals, more on that in a moment). Its proponents say it’s also more eco-friendly, producing less greenhouse gas emissions and requiring less water and land use than conventional meat. But price is a problem.
The price of cultured meat has gone down dramatically, up to the point where it is almost price competitive with meat; but almost is the key word. While estimates say lab-grown meat could be cost competitive by 2030, it’s not just there yet, and until it is, it’s unlikely to truly become mainstream. But new developments could change that.
The team from Singapore developed an unconventional method that uses magnetic pulses to stimulate the growth of cell-based meat.

Currently, lab-grown meat requires using other animal products — specifically something called “animal serum” — to feed the cells and get them to grow and develop. Animal serum is usually a mixture harvested from the blood of fetuses excised from pregnant cows slaughtered in the meat industry (something which happens often). You get much more meat than you put in (you can produce tons of meat for a few grams), but you still need a little bit to start with. Other methods to stimulate cellular production in lab-grown meat exist (particularly things like drugs or genetic engineering), but they are expensive and unlikely to be accepted by consumers.
This is where the new method comes in.
Alfredo Franco-Obregón from the National University of Singapore led a multidisciplinary team that uses a carefully-tuned pulsed magnetic field to culture myogenic stem cells, which are found in skeletal muscle and bone marrow tissue.
“In response to a short 10-minute exposure to the magnetic fields, the cells release a myriad of molecules that have regenerative, metabolic, anti-inflammatory and immunity-boosting properties. These substances are part of what is known as the muscle “secretome” (for secreted factors) and are necessary for the growth, survival and development of cells into tissues. We are very excited about the possibility that magnetically-stimulated secretome release may one day replace the need for FBS in the production of cultured meat,” the researcher said in a statement.
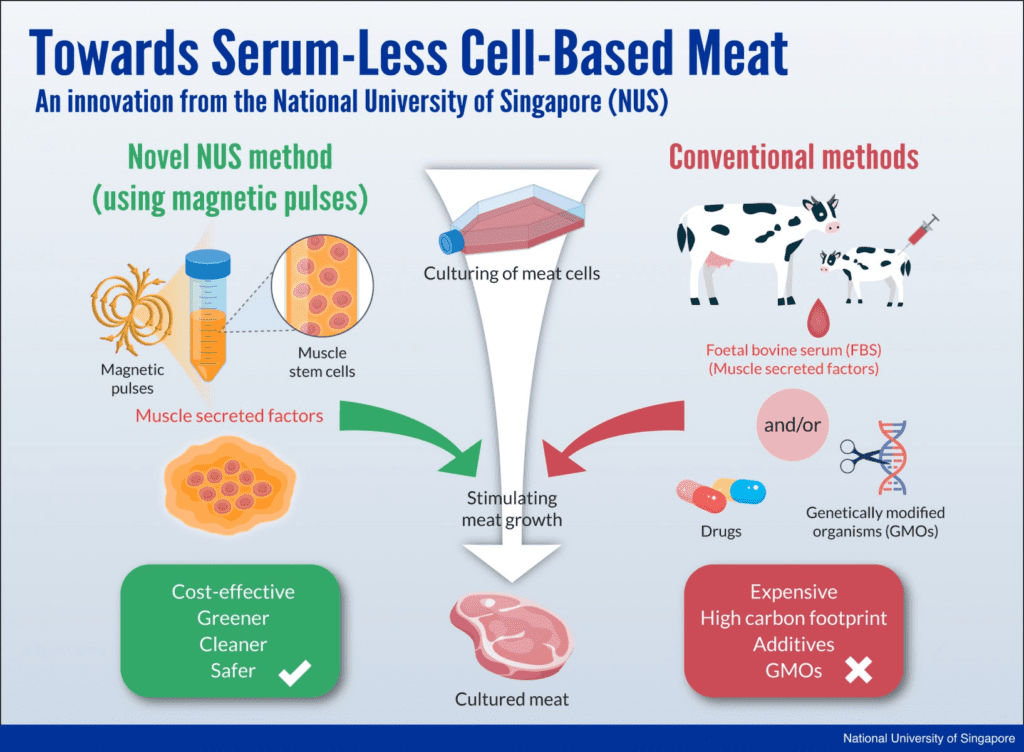
Not only is this process cruelty-free, but it is also cheaper than current practices as obtaining and using animal serun can be quite expensive.
“The growth-inducing secretomes can be harvested in the lab safely and conveniently, and also at low cost. This way, the myogenic stem cells will act as a sustainable and green bioreactor to produce the nutrients-rich secretomes for growing cell-based meat at scale for consumption. The muscle knows how to produce what it needs to grow and develop – it simply needs a little bit of encouragement when it is outside its owner. This is what our magnetic fields can provide,” the researcher adds.
The technique is also promising in medicine, the researchers note, as the same approach could be used to accelerate the recovery and growth of injured cells.
The researchers have published their findings in a peer-reviewed journal and have also filed for a patent for their new technology. They’re also looking to commercialize the method.
Was this helpful?



