In 2003, Dr. Lihong V. Wang at Washington University introduced one of the most exciting technologies we’ve had the chance to see in a long while. By combining ultrasound and light absorption, Wang and colleagues developed a new method that makes multicontrast images of biological tissues several inches below the skin. Among others, this allows specialists to see cancer in action with unprecedented detail and provide a faster and more accurate diagnosis. Recently, Want and colleagues at Washington University published a paper that refines this innovative optical imaging method by using what they call “time-reversal” technology that allows for better-focused light in tissue, such as muscles and organs.
Peering through the skin
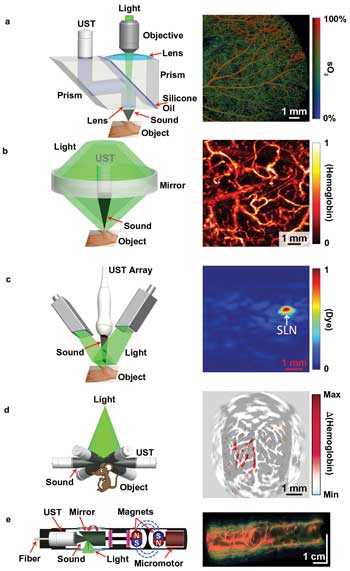
Called photoacoustic tomography (PAT), the technique achieves imaging depth by combining the spatial resolution of ultrasound with the high contrast resulting from light absorption by colored molecules such as hemoglobin or melanin. Typically, high-resolution optical imaging technology allows researchers to see about 1 millimeter deep into the body. PAT, however, can peer through biological matter at depths as big as 7cm!
What makes PAT really exciting is its ability to reveal the use of oxygen by tissues. When oxygen is burned in excess (hypermetabolism), this is usually a telltale sign that cancer is lurking. Diabetes also causes hypermetabolism. By using PAT, doctors can analyze patient tissue and provide an early-warning diagnosis without using a contrast agent – a big game changer!
The key to PAT’s success is converting light into sound waves, which scatter a thousand times less than light. A nanosecond-pulsar laser is used to this end, which is directed at the tissue. The thermoelastic expansion of the tissue causes the photons to convert into sound waves, later used to form images with a resolution associated with the ultrasound wavelength at tissue depths never before possible. Because scattering does not destroy photons, the technique makes it possible to reach a depth of about 7 cm.
Of course, there are many methods to examine living cells inside the body. Optical microscopy is used to examine organelles and cells, and nonoptical imaging techniques such as x-ray tomography are used for tissues and organs, but between these two domains – the macro and micro – a huge divide exists in scale. “Using the same contrast allows the same landmark features to appear in images acquired across length scales with different spatial resolutions,” Wang says.
With help from exogenous (introduced) contrast agents, PAT can image tissues such as lymph nodes that otherwise would blend with their surroundings. A huge improvement comes in sentinel node biopsy. Sentinel nodes are the nodes nearest a tumor, to which cancerous cells first migrate. To perform a biopsy, a surgeon injects a dye, a radioactive substance or both near a tumor. The body treats both substances as foreign, so they flow to the first draining node to be filtered and flushed from the body. Using Wang’s technique, however, a simple optical dye is injected that shows up clearly in photoacoustic images a vast improvement over its invasive counterpart.
Since 2012, the Washington University team has gone to great lengths to improve and refine their PAT tech. Reporting in the Nov. 2 issue of Nature Photonics Advance Online Edition, Wang and colleagues write how they are now using time-reversed adapted-perturbation (let’s call it TRAP for short) optical focusing to image movement in real time.

When light moves through stationary tissue it shows different than that that passes through moving tissue, like blood. The new refinement takes two successive images, then subtracts the stationary-moving light, retaining only the scattered light due to motion. Through a process called time-reversal, the light is then jettisoned back to its original sources where it becomes focused. This allows the PET technique to be a lot more efficient at tracking moving objects. Also, images can now be taken from greater depths.
Journal Reference: Cheng Ma, Xiao Xu, Yan Liu & Lihong V. Wang. Time-reversed adapted-perturbation (TRAP) optical focusing onto dynamic objects inside scattering media. Nature Photonics (2014) doi:10.1038/nphoton.2014.251
Was this helpful?



