In a novel research project, bioengineers at Duke University demonstrated how to grow functioning human muscles from induced pluripotent stem cells. It’s the first time scientists have shown that it’s possible to grow human muscles essentially starting from scratch, all by coaxing skin cells to generate stem cells, which ultimately turned into muscle tissue.
Previously, the same team grew functioning human muscle — the kind that contracts in response to a stimulus such as an electrical signal — in culture, starting with pea-sized globs of muscle, sourced from human volunteers.
While the previous attempts grew new muscle from native muscle, the present work is far more sophisticated since the resulting tissue doesn’t depend on donated muscle. What’s more, the ability to generate muscles starting from non-muscle tissue helps scientists grow far more of these cells. Bearing this in mind, the new technique will prove far more useful as far as genome editing and cellular therapies go. It also makes it possible to tailor custom treatments for rare muscle disorders such as muscular dystrophies or test substances for new drug discovery.
Skin to muscle
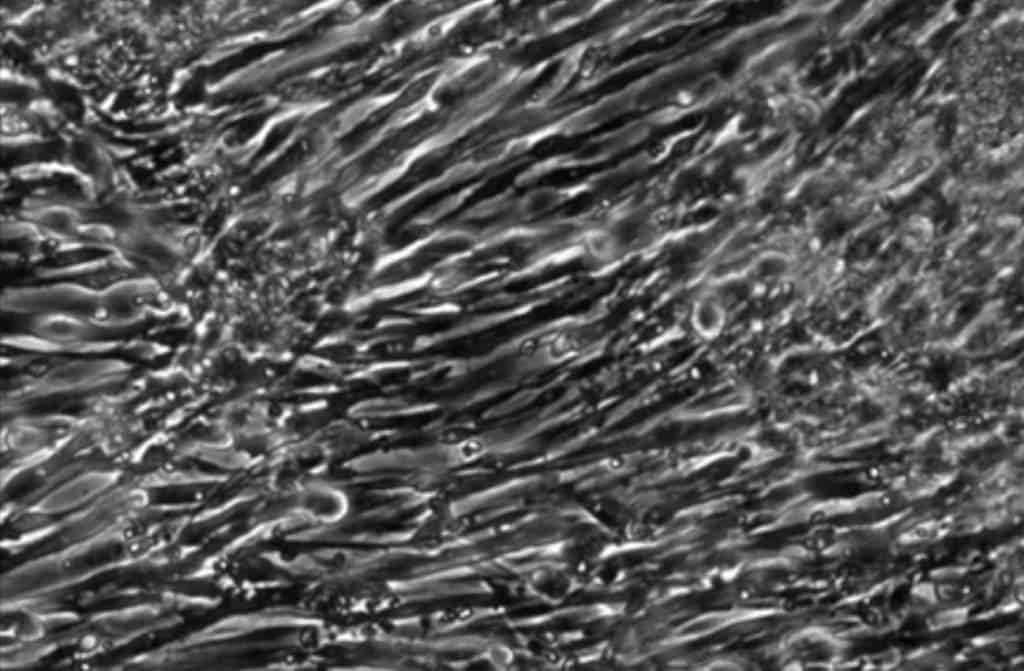
It all starts from human induced pluripotent stem cells (iPSCs), which are adult non-muscle tissues, such as skin or blood, that have been reprogrammed to revert to a primordial state. Just like any stem cells, iPSCs can then be programmed to differentiate into any kind of tissue. In our case, scientists flood the iPSCs with a signaling molecule called Pax7 which triggers muscle formation out of the cells.
“Starting with pluripotent stem cells that are not muscle cells, but can become all existing cells in our body, allows us to grow an unlimited number of myogenic progenitor cells,” explained Nenad Bursac, professor of biomedical engineering at Duke University. “These progenitor cells resemble adult muscle stem cells called ‘satellite cells’ that can theoretically grow an entire muscle starting from a single cell.”
It sounds easy enough when reading this but the truth is it took Bursac and colleagues years of trial and error before they got it just right. One important breakthrough that enabled cell proliferation into functioning skeletal muscle was the introduction of a unique cell culture conditions and 3-D matrix, rather than having to rely on the 2-D culture approaches that are typically used. This way, cells can grow and develop much faster and longer.
Once the cells reached a critical threshold, the scientists stopped the flow of Pax7 and started giving the cells the support and nourishment they needed to fully mature.
After two to four weeks of 3-D culture, the muscle cells aggregated into muscle fibers that can contract and react to electrical pulses and biochemical signals just like natural muscles would in response to neuronal inputs.
The lab-grown muscle fibers were put to the test when they were implanted into adult mice. Small ‘windows’ grafted on the backs of the mice allowed the researchers to observe how the muscles they’ve grown behaved inside the rodents. The muscles survived and functioned for at least three weeks, during which it progressively integrated with native tissue through vascularization.
The resulting muscle, unfortunately, is not nearly as strong as native muscle tissue nor that grown from muscle biopsies for that matter. It makes up for it, however, in versatility and the ability to grow many more cels from a smaller starting batch than other methods, such as the biopsy one. What’s more, the team’s new muscle seems capable of supplying a special reservoir of cells that muscles can use to regenerate themselves following injury or exercise — an important hallmark of natural muscle. Biopsied muscle tissue, in contrast, produces a far less richer reservoir.
“The prospect of studying rare diseases is especially exciting for us,” said Bursac. “When a child’s muscles are already withering away from something like Duchenne muscular dystrophy, it would not be ethical to take muscle samples from them and do further damage. But with this technique, we can just take a small sample of non-muscle tissue, like skin or blood, revert the obtained cells to a pluripotent state, and eventually grow an endless amount of functioning muscle fibers to test.”
Findings appeared in the journal Nature Communications.



