You hear about potential cancer treatments all the time, and despite significant and remarkable improvements, we’re still miles away from an actual cure for cancer; so what makes this therapy so great? Well… it just seems to work – on humans, suffering from cancer, not in a lab. For one woman, it seems to have completely dissolved a big tumor in just three weeks, and overall, 53% of patients experienced at least 80% tumor shrinkage.
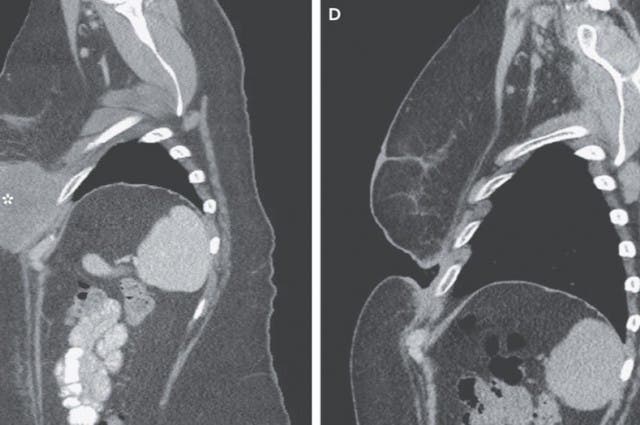
The patient is a 49-year-old woman who had a 4.2-mm ulcerated melanoma removed from her back 4 years ago. Five months ago, she had another subcutaneous nodule removed, this time under her left breast. However, there was persistent disease, and was at a high risk for further spread. After receiving this experimental treatment, her huge tumor completely disappeared, and left only a cavity behind, as you can see here. While most patients had significant improvements and over 20% were left without any visible tumors, doctors were simply amazed at the reduction in this case. In fact, it worked so good, that it got researchers worried about eliminating the tumor too fast:
“This patient had a rapid eradication of a large tumor mass after a single treatment with combination immunotherapy. We wish to bring this to the attention of our colleagues, not only as an example of the potential of immunotherapy to mediate dramatic and rapid antitumor effects, but also to point out a potential safety concern. Such an antitumor effect occurring in a transmural metastasis in the small bowel or myocardium, common sites of metastatic melanoma, could have grave consequences. It is ironic that we are now concerned about the possibility of overly vigorous antimelanoma responses.”
Alongside the obvious tumor in her chest, CT scans confirmed that the large tumor had been completely eradicated.
To make things even better, the therapy the scientists were investigating is FDA approved, involving two antibodies: Yervoy (ipilimumab) and Opdivo (nivolumab). They have very different types of action, but together they just seem to do the trick.
For the trial, researchers tested the drugs on 142 patients with melanoma that spread to other parts of the body or metastasized. The experiment followed what seemed to be the best standard for this type of study – comparing the results of standard treatment + placebo with the results of standard treatment + experimental treatment. In this case, patients were randomly assigned either Yervoy plus a placebo or Yervoy in combination with Opdivo.
“Patients were randomly assigned, in a 2:1 ratio and in a double-blinded manner, to receive both nivolumab and ipilimumab (combination group) or ipilimumab alone (ipilimumab-monotherapy group),” the published paper reads.
As mentioned above, over half of all the patients who got the combination experienced at least 80% tumor shrinkage, and melanoma became undetectable in 22% by the end of the study – spectacular figures for stage IV cancer.
Although the trial is now over, doctors obviously plan to include even more people in the future. You can read her case study here, and the full study here.






