Researchers at the University of California San Francisco have developed a drug that removes the molecular disguises of cancer cells.
“It’s cancer” is undoubtedly one of the most dreaded diagnoses any patient can receive today. And a large part of the dread this disease causes comes down to the fact that our bodies cannot deal with the tumors as they would with other diseases. Cancerous cells employ chemical disguises that fool our immune systems that they are not a threat, allowing them to develop unperturbed.
External avenues of treatment such as radiation- or chemotherapy can be employed against cancerous tumors. But, without the support of our bodies’ innate defensive systems, many cases of cancer are beyond the reach of external treatments.
New research at UC San Francisco hopes to fix that problem by painting huge targets on the backs of cancerous cells. Their approach involves pulling a mutated version of the protein KRAS to the surface of cancer cells, which flags them as targets for an immune response.
Un-stealthing
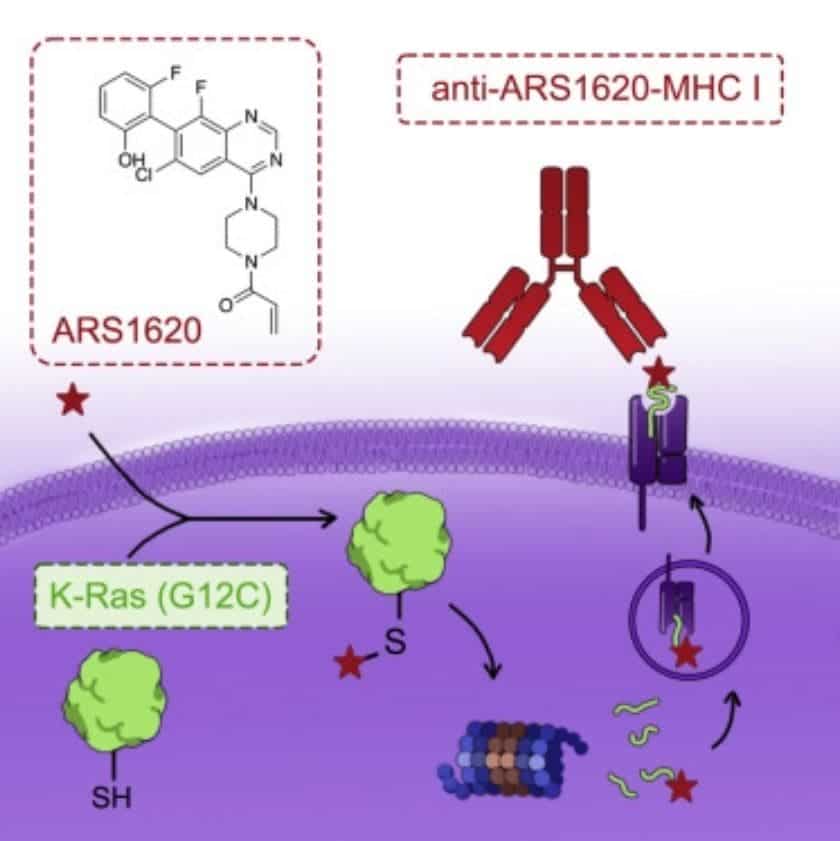
“The immune system already has the potential to recognize mutated KRAS, but it usually can’t find it very well. When we put this marker on the protein, it becomes much easier for the immune system,” said UCSF chemist and Howard Hughes Medical Institute Investigator Kevan Shokat, PhD, co-lead author of the paper.
Around one in four cancerous tumors have one kind of KRAS mutation, the team explains, which makes them one of the most common gene mutations in these cells. Given that such mutations are the target of sotorasib, a drug candidate which the Food and Drug Administration (FDA) has given preliminary approval for use in lung cancer, having the ability to bring the KRAS mutation to the fore would be invaluable in the treatment of cancers. In combination, these two substances could usher in a whole new approach to the treatment of cancers.
The researchers find the prospect of a new strategy that leverages our natural immune systems against cancers “exciting”. The findings, they add, could lead to better, longer-lasting treatments for cancer patients.
Immune cells read the proteins on the surface of particles they encounter in order to pick up on foreign bodies, unhealthy cells, or pathogens. Cancer cells have few proteins on the surface that are different from those of healthy cells. Most of the differences are contained inside the cells, where they are hidden from the immune system. The KRAS protein is the same, and for this reason, it was not considered to be a viable target for an anti-cancer drug despite how common it is in tumors.
The mutated version of KRAS drives the uncontrollable growth of cells in cancerous tumours. It is only slightly different from normal KRAS and its structure doesn’t allow drugs to readily bind to it. But work that Shokat carried out over the last decade described the protein’s structure and discovered a hidden pocket where a drug could bind to the molecule and block its activity. This research laid the foundation for the development and approval of sotorasib.
The current study set out to discover why sotorasib doesn’t work for all patients with KRAS mutations — in some it had no effect, while for others it caused the tumors to shrink first, and later become resistant to the drug and start growing again. Their goal was to discover whether they could devise a more reliable way of targeting KRAS.
They report that when a drug similar to sotorasib, ARS1620, targeted mutated KRAS, it doesn’t just block its role in promoting tumor development. It also allows immune cells to recognize the ARS1620-KRAS complex as a foreign and threatening molecule.
“This mutated protein is usually flying under the radar because it’s so similar to the healthy protein,” says Craik. “But when you attach this drug to it, it gets spotted right away.”
Although cancerous cells do develop uncontrollably, they still have many of the same safety systems in place as healthy cells do. Because they themselves recognize the ARS1620-KRAS complex as foreign, they push this molecule to the surface in order to signal the immune system for aid. This makes the molecule highly visible to immune cells, flagging cancerous cells as dangerous.
After finding a way to make KRAS visible, the team trawled through a library containing billions of human antibodies to identify those that could interact with this molecule. The most promising candidate of this step showed strong binding ability to ARS1620 alone and the ARS1620-KRAS complex in experiments with isolated protein and human cells.
Based on this antibody, the team engineered an immunotherapy procedure that enables T cells in our body to recognize the KRAS complex and engage with it. The team reports that this approach could kill tumor cells that contained mutated KRAS and were treated with ARS1620, including those that develop resistance to the effects of the drug itself.
“What we’ve shown here is proof of principle that a cell resistant to current drugs can be killed by our strategy,” says Shokat.
Before the treatment could be made available for patients, it will need to be tested in trials on animal and human models. Still, the greatest value of the research is that it establishes a whole new way of thinking about how to interact with dangerous cells inside out body.
The paper “A covalent inhibitor of K-Ras(G12C) induces MHC class I presentation of haptenated peptide neoepitopes targetable by immunotherapy” has been published in the journal Cancer Cell.
Was this helpful?



