
Everybody has used a thermometer at least once in their lives, but even without one, our bodies are decent sensors for measuring how hot or cold things are upon contact. We refer to this property as temperature which, in more technical terms, represents the average kinetic energy of the atoms and molecules comprising an object.
Heat or temperature?
Before we go any further with our discussion, it’s important to get something out of the way.
Often heat and temperature are used interchangeably — this is wrong. While the two concepts are related, temperature is distinct from heat.
Temperature describes the internet energy of a system, whereas heat refers to the energy transferred between two objects at different temperatures.
But, as you might have noticed, heat can be very useful when describing temperature.
Imagine a hot cup of coffee. Before pouring the hot elixir of life, the cup had the same temperature as the air surrounding it. However, once it came in contact with the liquid, heat was transferred, increasing its temperature. Now, if you touch the cup, you can feel that it’s hot.
But, given enough time, both the cup and its contents will reach thermal equilibrium with the ambient air. Essentially, they all have the same temperature, which is another way of saying there is no longer a net transfer of energy. Physicists call this the “zeroth law of thermodynamics”. By this principle, heat can only flow from a body that has a higher temperature than another body with which it is in contact — and never the other way around.
“Temperature is a thermodynamic concept characterizing the state of thermal equilibrium of a macroscopic system,” write physicists Bogdan F. Bogacz and Antoni T. Pędziwiatr. “Equality of temperatures is a necessary and sufficient condition for reaching thermal equilibrium.”
The dance of molecules
Everything in this universe is in motion, and motion begets kinetic energy. The faster a particle is moving, the more kinetic energy it has. In fact, kinetic energy increases exponentially with particle velocity.
Where does temperature fit into all of this? Well, temperature is simply an average measure of the kinetic energy for particles of matter. Another way of putting it would be that temperature simply describes the average vibration of particles.
Because the motion of all particles is random, they don’t all move at the same speed and in the same direction. Some bump into each other and transfer momentum, further increasing their motion. For this reason, not all particles that comprise an object will have the same kinetic energy.
In other words, when we measure an object’s temperature, we actually measure the average kinetic energy of all the particles in the object. However, it’s just an approximation.
Within this line of reasoning, the higher the temperature, the higher the motion of the particles. Conversely, when the temperature drops, the motion of the particles is slower. For instance, dyes spread faster through hot water than cold water. This also affects the phase of the substance. As NASA explains:
“When the state of matter changes its phase, the temperature and energy of matter also changes. At low temperature and energy we have a solid phase. At a medium temperature and energy we have a liquid phase, and at a high temperature and energy we have a gaseous phase.”
This is why at a temperature of absolute zero, the motion of particles grinds to a halt. Absolute zero is just a theoretical construct and, in practice, it can never be achieved. However, physicists have been able to cool things to a fraction of a degree above zero, trapping atoms and molecules, or creating exotic phases of matter such as the Bose-Einstein condensate (BEC).
It’s important to note that temperature isn’t dependent on the number of molecules involved. A boiling cup of water has the same temperature as a boiling pot of water — both containers have water molecules with the same average kinetic energy, regardless of the quantity of matter involved.
Temperature scales
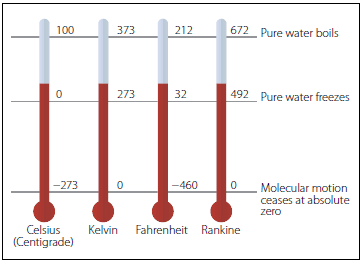
There are various scales used to describe temperature. In the United States, the most commonly used unit for temperature is Fahrenheit, while much of the rest of the world uses Celsius (or Centigrade). Physicists often prefer to measure temperature in Kelvin, which is also the standard international unit for temperature.
In nearly every country of the world the temperature scale used in everyday life is the Celsius scale. It is related to the Kelvin scale by a constant shift so that the increments in temperature have the same size but the zero of the scale is different,” writes physicist David Murdock.
For the Kelvin scale, zero refers to the absolute minimum temperature that matter can have, whereas in the Celsius scale, zero degrees is the temperature at which water freezes at a pressure of one atmosphere (273.15 Kelvin). At 100 degrees Celsius, water begins to boil at a pressure of one atmosphere, offering a neat, linear and relatable scale for describing temperature.
A worthy mention goes to the Rankine scale, which is most often used in engineering. The degree size is the same as the Fahrenheit degree, but the zero of the scale is absolute zero. Often just R for “Rankines” rather than °R is used for expressing Rankine temperatures. The zero of the Rankine scale is -459.67°F (absolute zero) and the freezing point of water is 491.67R.
How temperature is measured
Because of our innate ability to sense how hot or cold things are, humans have had little use for precise measurements of temperature throughout history. However, there have always been mavericks bent on learning about things just for the sake of unraveling nature or getting a kick out of doing science.
Hero, a Greek philosopher and mathematician, is credited with the idea for the first thermometer, writing in the 1st century CE about the relationship between temperature and the expansion of air in his work Pneumatics.
The ancient text survived the degradation of the Roman Empire and the dark ages that followed, until it resurfaced during the Renaissance.

It is believed that Hero’s work inspired Galileo Galilei to invent the first device that precisely measures temperature. The Galileo thermometer is composed of multiple glass spheres each filled with a colored liquid mixture that often contains alcohol but can even be simply water with food coloring added.
Each bubble has a metal tag attached to it that indicates temperature, which also serves as a calibrated counterweight that’s slightly different from the others. These floating balls sink or float inside the surrounding water sinking or climb up the water column slowly and gracefully. People still use them to this day, mostly for decorative purposes.
“A measure of temperature is obtained using a thermometer. Many physical properties of materials change sufficiently with temperature. Some such properties are used as the basis for constructing thermometers,” according to an NCERT physics textobook. “It is our common experience that most substances expand on heating and contract on cooling. A change in the temperature of a body causes change in its dimensions. The increase in the dimensions of a body due to the increase in its temperature is called thermal expansion.”
For more precise measurements, there’s the traditional mercury thermometer whose fluid expands at a known rate as it gets hotter and contracts as it gets cooler. It’s then just a matter of reading the measurement indicated by where the column of liquid ends on the scale.
Robert Fludd, an English physician, is credited with designing the first thermometer in 1638 that had a temperature scale built into the physical structure of the device. Daniel Fahrenheit designed the first mercury-based thermometer in 1714 that ultimately went on to become the gold standard of temperature measurement for centuries.
Was this helpful?



