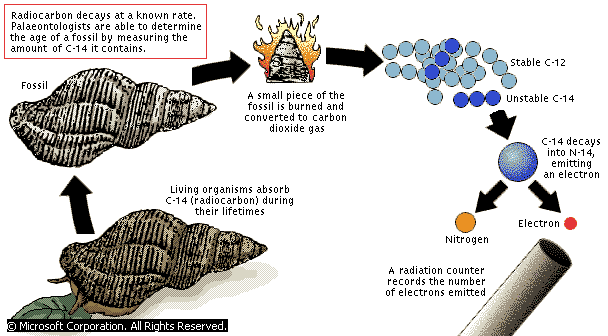
Scientists rely on a method called radiocarbon dating to determine the age of fossils or artifacts. With little or no other information available, the widely used method can accurately determine how old a sample is. This makes it one of the most powerful tools archaeologists, anthropologists and paleontologists have at their disposal. Rising CO2 levels in the atmosphere are, however, artificially aging the atmosphere and this might drastically interfere with the accuracy of radiocarbon dating. According to a new paper published by a team at the Imperial College London, “by 2050 a new T-shirt would have the same radiocarbon date as a robe worn by William the Conqueror a thousand years earlier.”
Radiocarbon dating works by comparing the three different isotopes of carbon, where an isotope is an element with the same number of protons, but different number of neutrons. So, essentially isotopes of the same element are chemically identical, but of different masses. While the lighter isotopes 12C and 13C are stable, the heaviest isotope 14C (radiocarbon) is radioactive, which means its unstable and decays. Over time 14C, which is naturally occurring and dispersed in the upper atmosphere, decays to nitrogen (14N). Over time 14C decays to nitrogen (14N).
Plants soak up the CO2 in the atmosphere through photosynthesis. In doing so, the plants ingest both 12C and 14C atoms. But eventually, animals who eat plants or animals who eat other animals who had eaten the plants also end up ingesting the 14C isotope, including you as a human. Thus, every plant and animal will have the same amount of 14C compared to 12C as the atmosphere (the 14C:12C ratio).
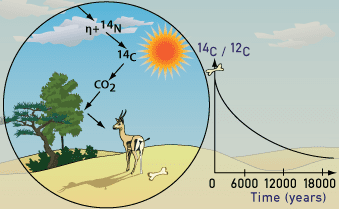
An accelerated dumping of CO2 into the atmosphere, especially since the 1970s, is beginning to mess up the accuracy of this radiocarbon dating. Fossil fuels are so old that they do not contain any trace of 14C, but when they burn up they flood the atmosphere with more 12C. Heather Graven, the atmospheric scientist at the Imperial College London, says he was surprised to find how much the emissions could “age” the atmosphere.
“If you think of parts of the deep ocean that are quite old, that have been sequestered for thousands of years, in the business-as-usual scenario, then the atmosphere would have the same radiocarbon fraction as the oldest part of the ocean,” she said. “It really is kind of backward. It’s very, very low.”
“Fossil fuel emissions are going to impact some of these neat applications, no matter what,” she said. “Now it’s a question of when it will happen and how low it will go.”
Radiocarbon dating could become significantly affected by 2020, if current emission trends continue.
“If we reduce emissions rapidly we might stay around a carbon age of 100 years in the atmosphere but if we strongly increase emissions we could get to an age of 1,000 years by 2050 and around 2,000 years by 2100,” Graven said.
Of course, there might be a way to account for these changes. If radiocarbon dating is to remain effective, the technique needs to be adjusted. After all, it would be quite the setback.