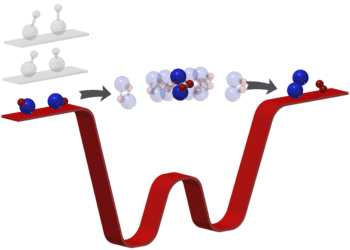
Chemical bonds – the bane of all high school students. Many see chemistry as an abstract way of describing the world, but for some chemists, it’s a very practical thing. Using a special type of microscopy, researchers triggered and visualized a chemical reaction at the atomic level.
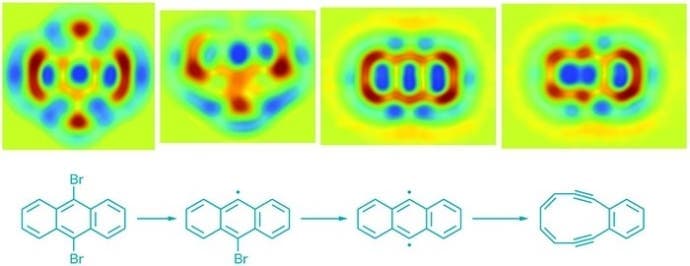
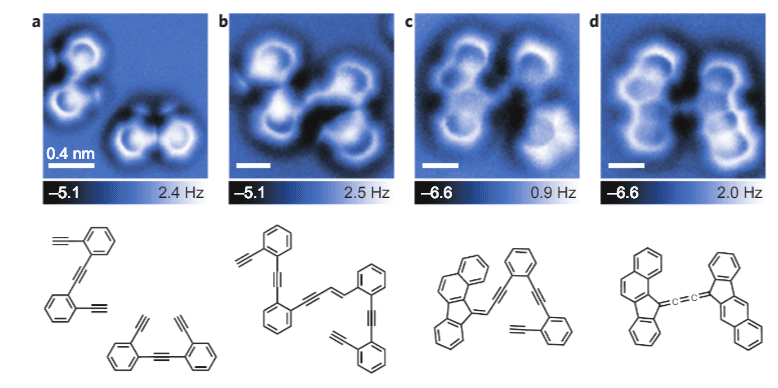
The team studied a version of the Bergman cyclization – an organic reaction and more specifically a rearrangement reaction taking place when an enediyne is heated in presence of a suitable hydrogen donor. Leo Gross of IBM Research Zurich and coworkers there and at the University of Santiago de Compostela used scanning tunneling microscopy (STM), a technique for nudging things at an atomic level taking advantage of a phenomenon called quantum tunneling. They then used atomic force microscopy (AFM) to image atomic-level details of that molecule as it formed. They managed to see its stages of formation as well as the final product.
The study “is a real breakthrough,” says Wolfram Sander of Ruhr University Bochum, a chemist who studies reaction intermediates. The ability to visualize and push the system in both reaction directions “is a great achievement,” he says.
The fact that they managed to both create and reverse is important. The technique could be applied to “initiate radical reactions by manipulating molecules at an atomic level” with potential applications in molecular electronics and subsequently electronic or medical devices.
Peter Chen of the Swiss Federal Institute of Technology (ETH) Zurich, also a reactive intermediates expert, notes that the technique also praised the results.
[It] “allows the chemist to initiate the reaction of a single molecule and then see the bonding changes in that very same molecule—not quite directly, but as close to directly as one can possibly imagine. This corresponds to the state of the art of what can be achieved” he said, referring to probe microscopy.
Journal Reference [open access]: Reversible Bergman cyclization by atomic manipulation.