Nematodes lack eyes, and why would they have any in the first place? Worms spend their lives inside the soil with little to no contact with sunlight. Yet, despite lacking eyes, worms do detect light — with their nose, sort of.

Inside the tissue of roundworms, researchers at the University of Michigan found a photoreceptor protein called LITE-1 that’s about 50 times more efficient at capturing light than rhodopsin — the photoreceptor protein found in the human eye. LITE-1 was first discovered among a family of taste receptors in invertebrates and it’s only the third type of photoreceptor found in animals.
“LITE-1 actually comes from a family of taste receptor proteins first discovered in insects,” said Shawn Xu, a faculty member of the U-M Life Sciences Institute, who is also a professor in the Department of Molecular and Integrative Physiology at the U-M Medical School. “These, however, are not the same taste receptors as in mammals.”
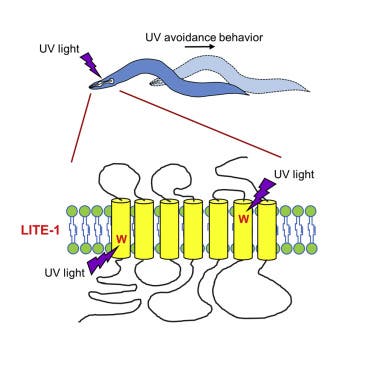
Xiu and colleagues were first hinted that the worms must somehow sense light after they flashed the eyeless, slithering critters and found these moved away. It could be that the nematodes sensed chemical reactions triggered by light interaction but eventually it was shown using spectrophotometric analysis that LITE-1 absorbs light. That’s strikingly different to the other two animal photoreceptors which react to photons (light particles) and do not simply absorb them.
“Photoreceptors convert light into a signal that the body can use,” Xu said. “LITE-1 is unusual in that it is extremely efficient at absorbing both UV-A and UV-B light—10 to 100 times greater than the two other types found in the animal kingdom: opsins and cryptochromes. The next step is to better understand why it has these amazing properties.”
That’s not all. Photoreceptors typically found in animals have two main components: a base protein and a light-absorbing chromophore. if you break the photoreceptors apart, the chromophore is still functional. When LITE-1 is broken into its constituents, however, the components’ ability to absorb is completely halted rather than simply diminished. Xu says this is proof that we’re dealing with a whole different photoreceptor model. The worm might be unique, at least in this regard. Perhaps other animals used LITE-1 but we’ve yet to found others.
Writing in the journal Cell, the researchers think LITE-1’s phenomenal ultraviolet absorption efficiency could make it a great ingredient for new, better sunscreens. Artificial photoreceptors used in sensors could also be developed based on the worm’s protein.
Not to worry, the sunscreen industry won’t have to farm millions of worms just so you can catch some rays in peace. Xu and colleagues found having the amino acid tryptophan in two places was critical to its function. When these residues were added to GUR-3, a taste protein from the same family, it began to strongly react to ultraviolet light with about a third the sensitivity to UV-B as LITE-1.
“This suggests scientists may be able to use similar techniques to genetically engineer other new photoreceptors,” Xu said.
Was this helpful?



