On a nanoscale luxurious track made of gold and silver, molecular machines competed in the world’s shortest race by distance. But although the track was a fraction of the width of a human hair, our molecular vehicles were even smaller which explains why the whole race took no fewer than 30 hours to designate the winner.
Not your typical drag race
The so-called “nanorace” took place in April at the CNRS’s Centre d’élaboration de matériaux et d’études structurales (CEMES) in Toulouse. This research facility houses a one of a kind scanning tunneling microscope (STM), which is equivalent to four STMs and without which such a race wouldn’t have been possible. An STM is a revolutionary instrument that uses a tip to measure the electric conductance between it and a material’s surface. This screening provides an electronic map of the surface down to the molecular or atomic level. Ultimately, the French STM could image tiny ‘vehicles’ made out of molecules at a resolution of 2 picometers, or 10-12 meters.
In this case, because the objects involved were extremely tiny, the STM instrument served a double role: measuring and propulsion. Each car had to be equipped with positive charge receptors while the microscope’s tip also supplied the energy to move the cars forward.

[panel style=”panel-warning” title=”The rules of the NanoRace at a glance” footer=””]
- The track is 20nm+1 turn+50nm+1 turn+20nm
- Maximum 38 hours of racing
- One section of the gold surface per team
- Maximum 6h to clean the track before the race starts
- No tip changes for 38 hours
- Pushing the NanoCar is not allowed
- Authorization to change the NanoCar
[/panel]
A tiny monster truck
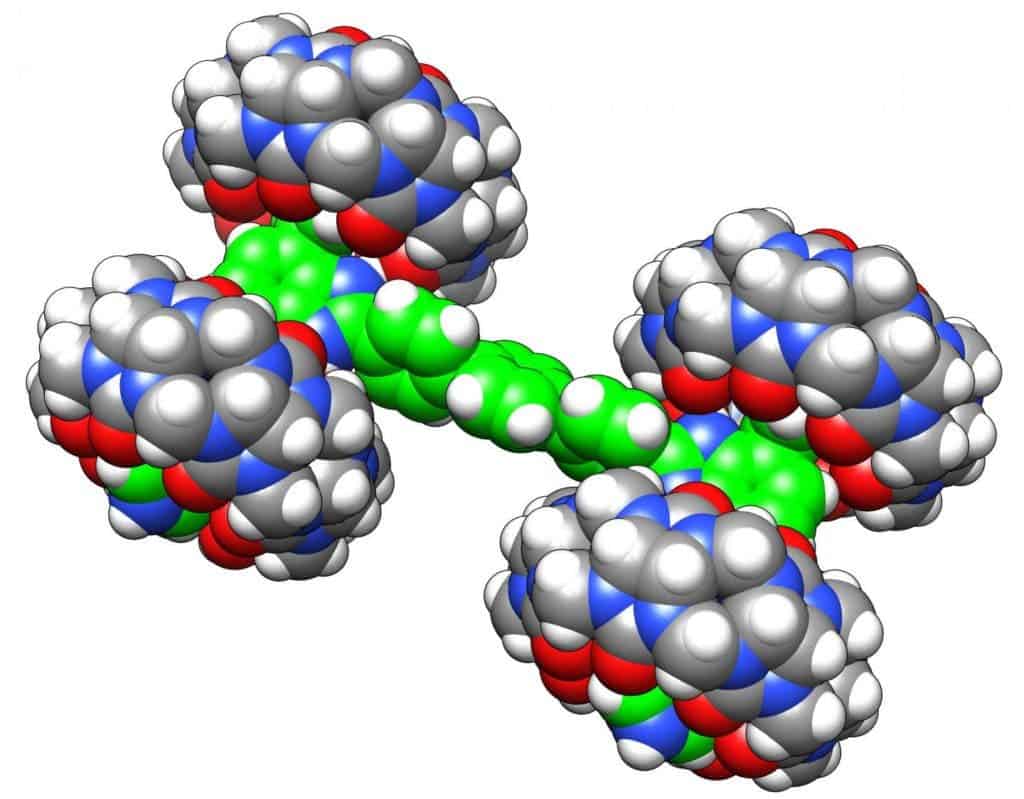
One of the teams involved in the race came from Ohio University. Their molecular contraption only measures 3.5 nanometers in length, made out of a chassis of H-shaped pseudorotaxane with four relatively large cucurbituril molecules serving as the wheels, as captioned in this article’s first image. Ironically, when it was completed, the molecular vehicle looked like a monster truck. Its official name is the Bobcat Nanowagon, though.
“The overarching goal was to advertise nanoscience to the public,” Eric Masson, one of the co-leaders of the Ohio University team said in a statement. “Then there was the technical challenge of manipulating multiple nanocars at the same time using a scanning tunneling microscope, or STM, instrument. Additionally, every team had its own goal. Ours was to see if we could deposit an intact supramolecular assembly onto a surface, and control its motion.”
In all, six teams from three continents entered the race meant to promote nanoscience to the general public. The Bobcat Nanowagon never left the team’s lab in Ohio, though, since the Toulouse facility only had four tips that could each control only one nanocar. Instead, the Ohio scientists left their nanowagon back home in the lab, but controlled it remotely from CNRS. “We manipulated the car at the atomic scale from half a globe away,” Saw-Wai Hla, who also co-led the Bobcat Nanowagon team.
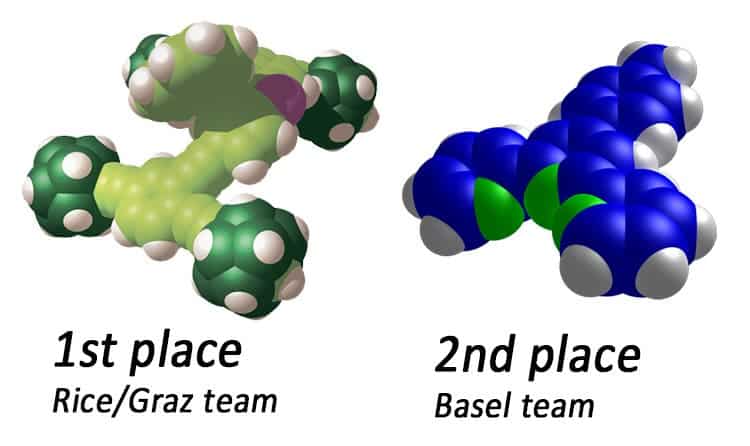
In the end, the Bobcat Nanowagon didn’t get to finish the race getting stuck 43 nanometers down the gold track after a Midwest power blip forced the molecules onto a rough section. It still traveled farther than three other contestants earning it third place in the first ever nanorace. First place went to the Nanoprix team, a US-Austrian collaboration (Rice/Graz), whose car traveled 1 micron at a snail’s pace of 29 hours. Second place went to a Swiss team from Basel which completed 133 nm of track in 6-and-a-half hours.
Besides promoting nanoscience to the public (and being extremely cool), such competitions could also provide new discoveries and insights. When Masson and Hla made their nanowagon, they had to make various iterations of the chassis molecules suspended in water. About 70 percent of the resulting structures looked like two-wheeled hover boards, a few had three wheels, and about 10 percent had all four wheels.
“That means that it was easier to break the chassis, a covalent bond, than to break the noncovalent bonds between the chassis and the wheels,” Masson said, who recently presented his team’s findings at the 254th National Meeting & Exposition of the American Chemical Society (ACS). “That’s completely counterintuitive because typically a noncovalent bond is much weaker than a covalent bond. It’s a theoretical curiosity.”
So there might be quite a few discoveries waiting to happen. At the end of the day, though, it’s all about the race itself, not the finish line.
“Our excitement is really about the fundamental science,” Hla says. “This is just the beginning.”



