
Though highly valued as one of the fuels of the future at the beginning of the century, the hydrogen economy has proven unfeasible or, maybe, ahead of its time. Today, the vast majority of hydrogen is produced through a highly energy-intensive process called methane reforming. This is expensive and carbon-intensive which defeats the purpose of green, alternative fuel. But by using a century-old idea, researchers from the Georgia Institute of Technology may have come up with a novel and efficient means to generate hydrogen.
Making hydrogen like a well-oiled machine
What Andrei Fedorov, a Georgia Institute of Technology professor, and his colleagues did was to adapt a four-stroke internal combustion engine so it churns out hydrogen gas with every cycle. Combustion no longer takes place in the cylinder. Instead, methane is reacted in the presence of a catalyst during the ‘power stroke’ to form hydrogen gas and CO2 as a byproduct. The carbon molecules are adsorbed internally so it can then later be recycled or stored to prevent it from entering the atmosphere.
Fedorov claims this method can be scaled to any size and could be used by anyone from household consumers to big industry to generate hydrogen on-site. Basically, anywhere where there’s a natural gas link.
“We already have a nationwide natural gas distribution infrastructure, so it’s much better to produce hydrogen at the point of use rather than trying to distribute it,” Fedorov said. “Our technology could produce this fuel of choice wherever natural gas is available, which could resolve one of the major challenges with the hydrogen economy.”
The hydrogen reactor works almost exactly like a four-stroke internal combustion engine with some notable distinctions. Natural gas enters through a valve that open when the piston is lowered and closes when the piston rises. During a conventional power stroke, the vaporized liquid fuel is ignited by a spark plug if we’re talking about a gasoline-powered IC or by spontaneous combustion if this is diesel. In the case of the hydrogen four-stroke reactor, the power stroke raises the pressure of the natural gas causing it to reach 400 degrees Celsius. At this temperature, the gas reacts with water in the presence of a catalyst forming hydrogen and CO2.
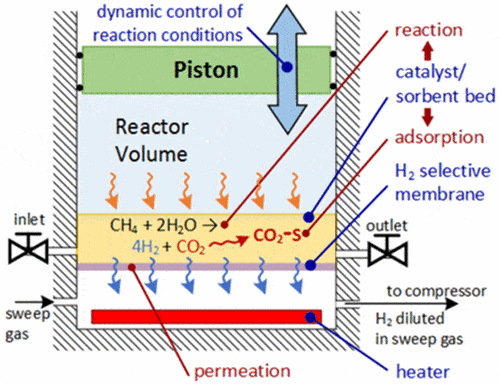
The molecular H2 exits the reactor through a selective membrane while the pressurized CO2 is adsorbed by a sorbent material, mixed with the catalyst. Once H2 exits, the piston is lowered, reducing the pressure inside the cylinder so the CO2 is released from the sorbent material into the cylinder. The piston rises again for its final stroke to expel the CO2 and clear the reactor for a fresh new cycle.
This reactor, aptly named CO2/H2 Active Membrane Piston (CHAMP) reactor, uses only two molecules of water for every molecule of methane to produce four hydrogen molecules, and operates at between 400 and 500 degrees Celsius. Conventional methane reforming needs three water molecules to make four hydrogen molecules and requires steam heated at 900 degrees Celsius.
CHAMP’s cylinder volume can be adjusted to virtually any scale to produce anywhere from a few kilograms of H2 to hundreds on a daily basis.
“All of the pieces of the puzzle have come together,” said Fedorov, a professor in Georgia Tech’s George W. Woodruff School of Mechanical Engineering. “The challenges ahead are primarily economic in nature. Our next step would be to build a pilot-scale CHAMP reactor.”
“We took the conventional chemical processing plant and created an analog using the magnificent machinery of the internal combustion engine,” Fedorov said. “The reactor is scalable and modular, so you could have one module or a hundred of modules depending on how much hydrogen you needed. The processes for reforming fuel, purifying hydrogen and capturing carbon dioxide emission are all combined into one compact system.”
The machine was described in the journal Industrial & Engineering Chemistry Research.
Was this helpful?



