Physicists at Penn State University are experimenting with a novel type of refrigerator. No bigger than a beverage coaster, this so-called electrocaloric refrigerator generates a temperature difference of about 2 degrees Kelvin between its hot and cold ends. The device is based on the electrocaloric effect which involves switching electric fields to a material to increase or decrease its temperature. Though the absolute cooling effect might not sound impressive at this point, scientists say this method has the potential to be more energy efficient than conventional cooling methods employed today in modern refrigerators or other cooling devices.
Unlike air conditioners and refrigerators, most of which are based on traditional vapor-compression-cycle-based cooling, the electrocaloric refrigerator is based on a reversible temperature change under an applied electric field. It is often regarded as the reverse of the pyroelectric effect.
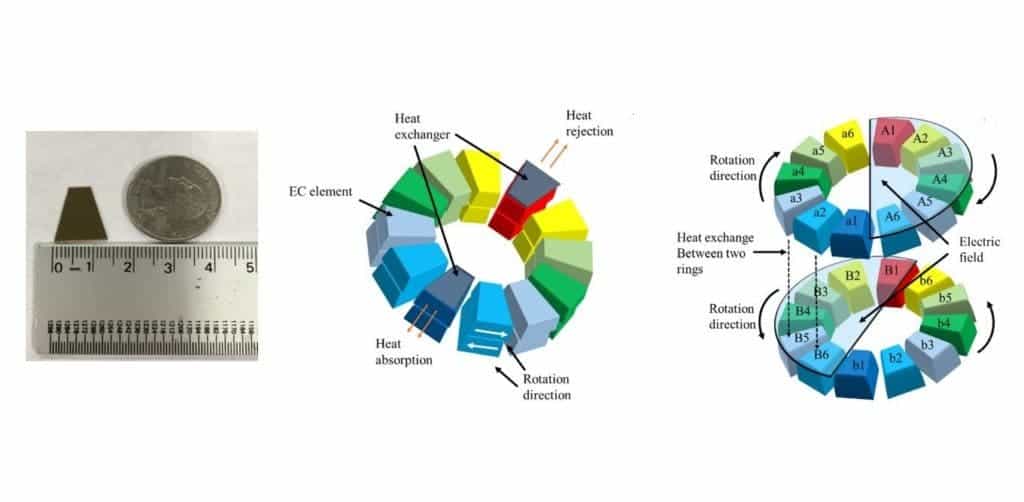
The Penn State team led by Professor Qiming Zhang experimented with electrocaloric materials and arrangements. Their device consists of multiple ceramic rings each made up of around a dozen or so coin-sized elements. These rings rotate at a rate of several RPMs and when the elements of a ring rotate toward the hot end, an electric field is applied to the elements ejecting heat in the process. When the elements rotate toward the cool end, the reverse process occurs — the electric field is turned down to zero turning the elements into heat absorbers. Because heat is exchanged between adjacent rings that rotate in opposite directions, the cool end of the device cooler and the hot end hotter over time.
Previously, the researchers simulate what their device might behave like and so it was quite the victory for them to see their novel refrigerator working as expected. The prototype currently maintains a temperature difference of 2 K between the hot and cold ends. It’s made of commercially available ceramics and only two electrocaloric rings which can only mean there’s a lot of room to augment performance.
In March 2006, Zhang and colleagues made an electrocaloric refrigerator from thin films of PZT (a mixture of lead, titanium, oxygen, and zirconium) the strongest electrocaloric response yet reported, with the materials cooling down by as much as ~12 K (12 °C). However, the effort required an electric field change of 480 kV/cm and at an ambient temperature of 220 °C (430 °F).
“In the future, we will focus on developing electrocaloric materials (including polymer and ceramic) which can generate the electrocaloric effect at very low voltage,” Zhang told Phys.org. “We will also work on scaling up the current state-of-the-art electrocaloric materials to the commercial scale, which can be used reliably with an applied voltage of less than 200 volts.”
Hopefully, an electrocaloric refrigerator will one day become practically viable to consumers as more energy-efficient and lightweight alternative to today’s fridges. These could be anything from wine chillers to computer coolers to air conditioners.
Journal reference: Tian Zhang et al. “An electrocaloric refrigerator with direct solid to solid regeneration.” Applied Physics Letters. DOI: 10.1063/1.4986508.


