
A study published in the journal BMC Biology by researchers from the United Kingdom, China, and Germany revealed the discovery of nanobodies akin to each other that inhibit the fibril formation of alpha-synuclein, the protein responsible for Parkinson’s Disease (PD).
Parkinson’s Disease, the degenerating movement disorder where nerve cells become impaired and die, disrupts most aspects of life for those stricken with it. The NIH describes this affliction as sporadic, progressive, and chronic. It is thought to result from genetic mutation and environmental factors.
The telltale sign indicating its presence is easily discernible: the presence of Lewy bodies in neurons. Difficulty walking, tremors, and rigidity are early symptoms. Later stages include severe cognitive decline. Presently, PD has no cure.
How PD works
Alpha-synuclein proteins form Lewy bodies then conjoin into cytotoxic oligomers, and finally, aggregate into fibrils (small fibers).
[accordion style=”info”][accordion_item title=”What are those?”]Oligomers are long chemical strands, made up monomers. Unlike plastics, which are polymers and can theoretically be any length, oligomers only contain a few monomers, i.e. they are shorter. “Cytotoxic” means these substances are toxic to life.[/accordion_item][/accordion]
They kill dopamine-producing neurons near the base of the brain, resulting in a loss of this chemical messenger molecule — among others, it handles signaling between the substantia nigra and the corpus striatum. This loss leads to neurons with abnormal firing patterns and uncoordinated movement. To illustrate the severity of this illness, most people with Parkinson’s have a noticeable absence of dopamine-producing cells in their substantia nigra. PD victims lose 60-80% of these cells.
Scientists seeking a remedy for this immutable aberration tried a new pair of nanobodies on alpha-synuclein. Derived from the antibodies of camels, they work remarkably in the laboratory. Their names: NBSyn2 and NBSyn87.
They exceed the abilities of previously studied antibodies and nanobodies. Whereas those took a while to cause sufficient changes to oligomer concentrations, these carry out a rapid conformational conversion. This is great news, as passive immunization using antibodies targeting alpha-synuclein has shown promise in several clinical trials.
After studying them, scientists outlined each of their strengths and shared esoteric knowledge on the nanobodies. To allude to them, NBSyn2 is better at reducing cytotoxicity, they explain, while NBSyn87 slows fibril formation to a greater degree. They break it down further, stating NBSyn2 attributes its ability to its lower positive charge and decreased interaction with the cell membrane. NBSyn87 achieves its feat by binding with a higher affinity, closer to the folding region of the alpha-synuclein protein, thereby slowing fibril formation with steric hindrance.
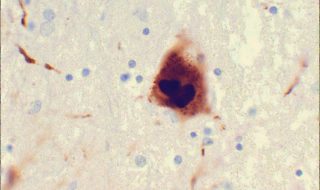
Working in tandem, these nanobodies do three things: First, they prevent alpha-synuclein from aggregating into Lewy bodies; second, they hinder it from propagating in a prion-like manner; third, they destabilize cytotoxic oligomers through conformational conversion, antagonistically altering its final make-up and stopping its maturity.
Nanobodies got their name, unsurprisingly, from being miniature in size. They’re almost 10 times smaller than normal antibodies. And, being comprised of only heavy chains, they can easily cross the blood-brain barrier and bind to a variety of “hard to reach” epitopes.
[accordion style=”info”][accordion_item title=”Epitope”]An epitope is the part of a molecule to which an antibody ties — this bonding is how they ‘cure things’.[/accordion_item][/accordion]
Only 0.3% of brain synapses use dopamine; however, it has an important role to play in movement, pleasure-seeking, avoiding bad situations, and addictive behaviors, among others. Clinically, dopamine is used to dilate the renal artery and increase cardiac output. It’s in the same family of substances as epinephrine, norepinephrine, histamine, and serotonin — a family called catecholamines.
Mostly affecting those 60 years old and older, an estimated 7 to 10 million people worldwide have PD. That’s 0.09% to 0.13% globally, with the current population standing at 7.6 billion people. In 2009, the highest prevalence of this disease was in the Northeastern United States’ Amish community.
Years of research aimed at developing a quick-acting treatment for PD, using full-length antibodies and antibody fragments, targeted against different regions and different species of alpha-synuclein, led to this study. Still afoot, this undertaking is going somewhere. Now, researchers await the animal testing phase of these nanobodies with anticipation. “This find has the potential to form the basis of a new therapeutic strategy to combat PD and related protein misfolding conditions,” state researchers.
In time, other major protein misfolding diseases that these nanobodies might develop into an antidote to treat include Alzheimer’s Disease, Huntington’s disease, Creutzfeldt-Jakob disease, Cystic Fibrosis, and Gaucher’s Disease.
Was this helpful?



