After graphene proved to be one of the greatest discovery of the century, material scientists became inspired to see if other 2D meshes (just one atom thick layer of material) could be made from other elements. In time, we’ve heared about silicene, phosphorene or germanene. Now, a group from China reports for the first time stanene: a honeycomb 2D arrangement of tin (Sn) atoms, with a a bismuth telluride support that buckles the whole structure. Stanene is extremely exciting because it’s been previously theorized that it could transfer electricity without heat loss, implying huge energy savings and increased performance for semiconductor applications.
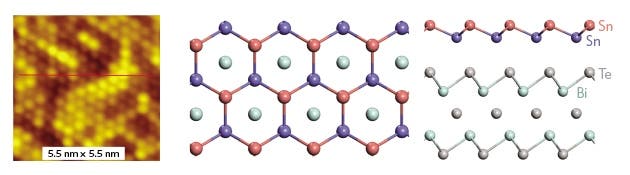
The zero energy loss of stanene was predicted by Stanford researchers in 2011. That’s because stanene is a topological insulator: it can’t conduct electricity inside the bulk material, but it can at the edge.
When electrons travel through a material they bump into atoms, transferring some of the energy to the atoms that causes them to jiggle. This extra vibration gives off energy we know as heat. But in order for the electron (quantum particle) to collide with an atom, it needs to fill an unoccupied quantum state with the correct spin, energy and momentum. Because there are fewer collisions in an topological insulator, such a material used as a conductor could save a lot of energy. However, Shou-Cheng Zhang, a physicist at Stanford University in California and co-author of the present study, says that his team was yet to prove that stanene can conduct electricity without generating any waste heat.
What they have, for now, is prove that stanene can exist in this state. The researchers made the mesh by vaporizing tin in a vacuum and allowing the atoms to waft onto a supporting surface made of bismuth telluride. This allowed 2D crystals to form, but the bismuth telluride also interacts with the mesh causing the material to lose its topological insulating properties. There’s another possible shortcoming. Theory says stanene should like a buckled honeycomb, but the researchers could only see see the upper ridge of atoms with their scanning tunnelling microscope, as reported in Nature Materials. To be sure of its properties, the researchers should make pure stanene, and not some other tin arrangement.
Still, efforts so far with graphene and its cousins have proven to be extremely lucrative and, most of all, promising. “It’s like going to the Moon,” says And Guy Le Lay, a physicist at Aix-Marseille University in France. “The first step is the crucial step.”
Was this helpful?



