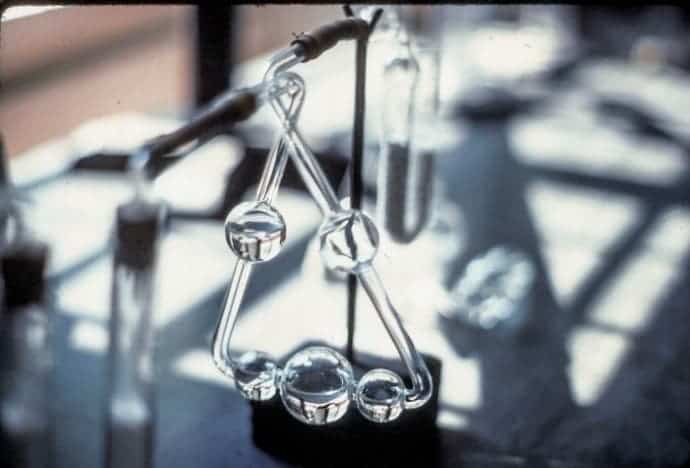
This fashionable triangle-shaped glassware isn’t an office decoration, but a true vestige of the early analytical chemistry. It’s called the kaliapparat, a hollow glass tubular device used to measure carbon content in substances in 1830 by German chemist Justus Liebig, widely considered the father of organic chemistry.After it first appeared, because it was so reliable, it spread throughout labs in Europe and North Africa within a matter of years. Look at this device again. Does it look familiar? It’s none other than the American Chemical Society’s logo – the same logo still in use today more than a century later. As such, the kaliapparat is one of the most important chemistry vessel and analytical tool in history. It’s also one of the most obscure. Few chemists know about it. Most ACS members have no clue what it is or what the logo means for that matter.

The kaliapparat’s working principle is very simple. Inside a separate glassware a substance is burned, and the fumes carrying the combustion products are directed through the kaliappart. First, the products pass through calcium chloride that absorbs the water vapor. Then, the combustion train, devoid of water, goes through the kaliapparat itself where a solution of potassium hydroxide is placed. The potassium hydroxide absorbs the CO2 combusted from the substance.
The potassium hydroxide is held inside the bottom three bulbs. If there’s violent bubbling inside the bulbs, it means the CO2 absorption process is still taking place. At the third bulb, there should be little to no absorption going on. Simply ‘by eye’, you have a great status of how your experiment is working.
When the operation is over and the product is fully burned, a chemist simply has to weigh the hydroxide sample. By subtracting post and pre-combustion mass, you get the mass of the CO2. Since we know the mass of oxygen, we can tell how much carbon a sample weighs.
Today, samples are also burned to determine their carbon content, but the instruments are far more sophisticated. Pure oxygen is combusted with the sample, then products are accelerated, passed through filters and read with atomic accuracy by infrared scanners and spectrometers. Yet the kaliapparat is still competitive, and great for estimates. What it lacks in sophistication, it makes up for in innovation.
In 2005, Alan Rocke, a historian of chemistry at Case Western Reserve University, made a study in which he put Liebig’s kaliapparat experiments to scrutiny. “Some historians thought the kaliapparat mythology was overplayed,” Rocke says, “and there was more than a little bit of uncertainty about what the device could actually do.”
They tested the kaliapparat on tartaric acid, urea, cinchonine, and narcotine, they found that the analytical method was remarkably accurate. “It’s mind-boggling that the simple apparatus can give you the quality of modern analyses.” Of course, there were some mishaps as well. The kaliappart proved unreliable for nitrogen. It also can’t analyze complex alkaloids such as morphine or quinine. Liebig knew this and eventually instructed his students to introduce synthesis as a method for investigating organic chemistry. You can say that inspired by the kaliappart, or its limitations to be more precise, synthetic organic chemistry was born.

“The image of a chemist, wearing a lab coat, in front of a bubbling glass apparatus goes back to the kaliapparat,” says William Brock, an emeritus professor at England’s University of Leicester and a Liebig biographer. “It became a symbol, like the periodic table, for what chemists do.”
Liebig’s Kaliapparat remained in use for three quarters of a century and became a badge of honor for organic chemistry. It’s also a physical badge appearing in the logo of the American Chemical Society, on the southwest corner of Sterling Chemistry Laboratory, and as a woodcarving in Yale’s Chemistry Library.
Was this helpful?



