The future of antivenom production might finally be here, and it couldn’t come soon enough.
Snakebites might just be the biggest health problems you’ve never heard about. It’s virtually inexistent in many parts of the world but takes a massive important toll in other parts.
Snakebites kill a whopping 81,000 people a year, disabling over 400,000. If you’re reading this, the odds are that you’re not really subjected to this risk, as the vast majority of problems occur in sub-Saharan Africa or rural parts of Asia where medical facilities are few and far between.
Nevertheless, the impact of snakebites is substantial, and it is growing. The fact that antivenom is so expensive is not helping the cause: the cheapest antivenom vial sells for around $10, but the price can vary dramatically. In the US, a vial can go for as much as $2,300 per dose, and even more in extreme cases. Unsurprisingly, antivenom is often unavailable or unaffordable exactly in the areas that need it most.
Antivenom production has changed surprisingly little in the past century. It was first synthesized in the 1800s when Albert Calmette, a French physician working in Brazil, witnessed a startling number of patients suffering from snakebites. His approach was similar to that of a vaccine, instead of directly injecting the human, he first injected small amounts of venom into the body of animals, and then extracted a hyperimmunized serum to inject into humans.
“Milking” the snakes was done by hand, a rudimentary and challenging process, and is one of the main reasons why antivenom is so expensive.
This method is still used today, although more than a century has passed.
But three PhD students working in the group of Hans Clevers at the Hubrecht Institute in Utrecht, the Netherlands, might change that. They were inspired by their colleagues who had developed human organoids and wondered if the same thing could be done with reptile organoids.
An organoid is a miniaturized and simplified version of an organ produced in vitro in three dimensions that shows realistic micro-anatomy and can perform organ functions.
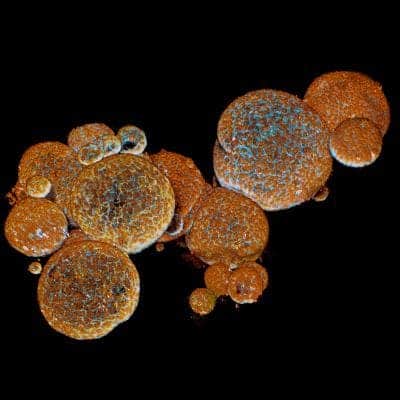
They collected venom gland samples from 9 snake species, attempting to build miniaturized versions of these in a Petri dish. After a bit of process tweaking, not only did it work — but the process was surprisingly similar to that of growing human organoids.
“The similarity between the growth conditions for human and snake tissues was staggering, with the main difference being the temperature”, says Jens Puschhof (Hubrecht Institute). Since the body temperature of snakes is lower than that of humans, the venom gland organoids only grew at lower temperatures; 32ºC instead of 37ºC.
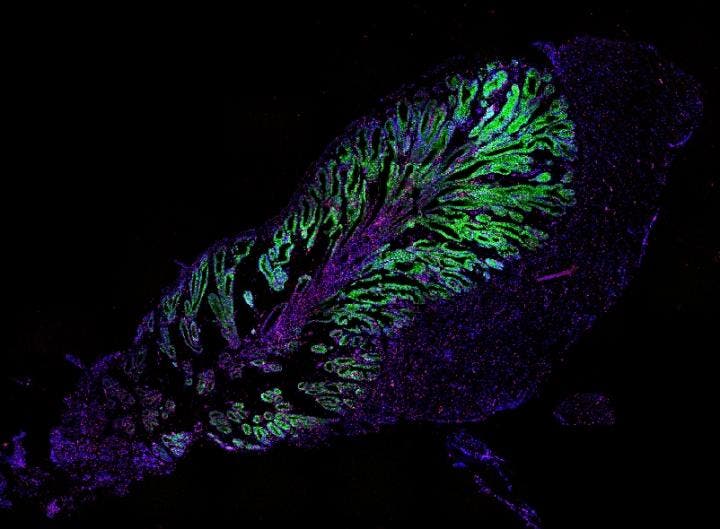
They then used a high-resolution microscope to study what these organoids were really doing. They found that the organoids were filled with structures that resemble venom.
Researchers could control the composition of the venom, changing key components — much like neurotoxins in different snake species differs. They could also direct the growth of the organoids
“We know from other secretory systems such as the pancreas and intestine that specialized cell types make subsets of hormones. Now we saw for the first time that this is also the case for the toxins produced by snake venom gland cells”, explains Joep Beumer (Hubrecht Institute).
The findings could open a new age in antivenom production, as well as help develop new venom-based treatments. At the very least, this could help drive the price of antivenom down, though the possibilities could be much more far-reaching.
In addition, this suggests that organoids of other animals could be developed in the lab, something which could have important implications for healthcare and beyond.
Researchers are now working to set up a large collection of such organoids, starting from 50 toxic reptilians. Yorick Post, one of the study authors, concludes:
“It was amazing to see that what started with our curiosity about potential snake venom gland organoids transformed into a technology with many potential applications affecting human healthcare”
The study has been published in the journal Cell.



