In some circles, iboga — a potent psychedelic drug obtained from the root bark of the African plant Tabernanthe iboga — is hailed as a miracle substance capable of instantly eliminating cravings and withdrawal symptoms for even the most heavily addicted opioid users. But iboga is a potent drug in itself that induces intense hallucinations and potentially deadly side effects. Anecdotal evidence of iboga’s therapeutic effects for tackling drug abuse and addiction is abundant, but what does the science actually say?
What is iboga

Ibogaine (12-methoxyibogamine) is the main alkaloid of at least 12 alkaloids found in the Tabernanthe iboga plant. The substance seems to activate the glial cell line-derived neurotrophic factor (GDNF) pathway in the ventral tegmental area of the brain, increasing its expression. Previous studies have found that the GDNF pathway plays a unique role in mitigating the negative effects of drugs with a high potential for abuse and raises the survivability of adult dopamine-related neurons. The synaptic remodeling may change the responsiveness of the mesolimbic dopaminergic system, thereby canceling the ‘rewarding effect’ and neuroadaptations induced by drug abuse. Such ibogaine-induced changes in GDNF expression may serve to explain the psychedelic drug’s potential efficacy against serious drug addiction.
Does ibogaine work?
The United States is facing a dramatic opioid crisis, with thousands of Americans getting killed annually from overdosing on prescription opioid drugs but also illegal opioids such as heroin. The country has only 5% of the global population, yet consumes 80% of the world’s pharmaceutical opioids.
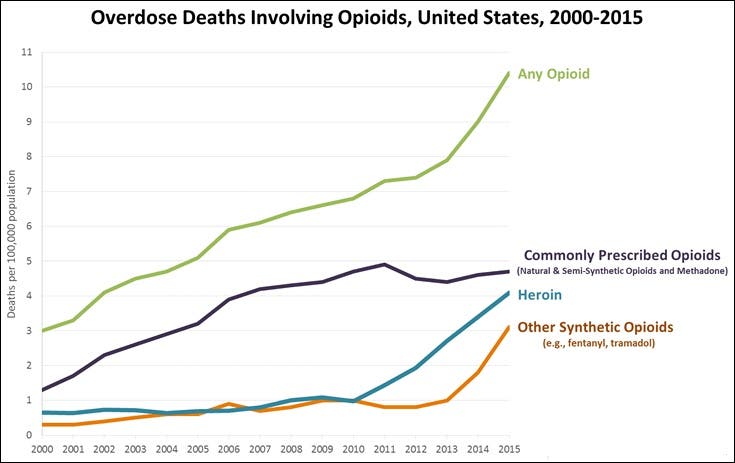
More and more Americans seem to succumb to opioid abuse, a problem exacerbated by a lack of effective treatments against morphine, heroin, methadone, and oxycontin addiction — and this is where iboga might make a difference. According to anecdotal evidence, a single dose is capable of alleviating drug craving and relapse of drug use for a period of time of weeks to months.
Scientists have been aware for decades of the anecdotal therapeutic effects of iboga but research has always been difficult given the drug’s classification as a Schedule I substance, up there with heroin. It is similarly scheduled in 9 of the 28 countries presently in the European Union. However, New Zealand, Brazil, and South Africa have classified ibogaine as a pharmaceutical substance and restrict its use to licensed medical practitioners. Fortunately, some scientists have been allowed to work with iboga and have published their results.
Animal studies suggest that within 24 hours after ingesting ibogaine, the alkaloids produce significant attenuation of opioid withdrawal signs in different animal species. What’s more, the substance also reduces the self-administration of other potent drugs such as cocaine, amphetamine, methamphetamine, alcohol, and nicotine.
In one study performed on humans, involving 33 participants with opioid dependence, withdrawal signs were completely resolved in 29 of the participants (88%). This was not a controlled clinical trial, however.
In a follow-up study conducted in 2004, researchers found that 67% of the 21 participants ceased the use of opioid drugs. The rest (33%) did not end the use of their primary or secondary drugs of abuse but decreased the amount of drug use nevertheless. The overall average drug-free period of all participants was 21.8 months — the median, however, was of 6 months. In another more recent follow-up study published in 2017, which involved 30 participants, 15 subjects (50%) reported no opioid use during the previous 30 days — an effect that “was sustained up to 12 months in a subgroup of subjects.” What’s important to note is that “ibogaine appears to have had a clinical effect in some subjects with histories of failure of other treatments,” the researchers wrote.
The psychological effects of ibogaine
An ibogaine treatment will induce a very intense trip, which has earned it an important place in medicinal and ritual practice in the African spiritual traditions of the Bwiti tribe in the Congo basin of Africa.
The iboga trip can be described in three phases:
- In the first phase (0-1 hour), the alkaloids gradually cause changes in visual, auditory, and body perception. Physical symptoms include dry mouth and difficulty in coordination.
- The second phase (1-10 hours) is often described as a sort of ‘waking dream’ where the patient undergoes intense visual hallucinations, a buzzing sound, and changes in the perception of time, space, and their own body. Nausea is a common symptom during this phase and patients report feeling physically heavy. The ibogaine visualization during this phase, which occurs with the eyes closed, can be either realistic or symbolic. Testimonies included visions of exotic cultures, deceased people, memories from childhood, or bizarre adventures through one’s own brain or DNA. The hallucinations tend to cease if the patients open their eyes. This phase usually ends abruptly.
- The third phase (11-36 hours) is regarded as a period of deep introspection. During this phase, patients will generally review memories, often concerning traumatic or emotional experiences, personal relationships, and important life choices. Subjects may find themselves pondering earlier experiences in life, imagining alternatives to their choices. The experience may result in a more responsible attitude towards future life choices but also forgiveness towards oneself and others for past transgressions.
Ibogaine safety
Worldwide, there are more than 3,000 private clinics and retreats where iboga has been established as a “medical subculture.” Thousands of people have so far taken iboga, largely in an unsupervised, non-medical setting (not in the formal sense of the word), with many anecdotally reporting overcoming addiction.
It’s worth noting however that iboga treatment is not without perils. Common physiological side effects during the first phase include nausea, mild tremor, and ataxia. In fact, some people have died after taking iboga, apparently due to a history of other medical conditions.
Between 1990 and 2008, 19 individuals died within 1.5–76 h of taking ibogaine. In most cases, the deceased had comorbidities and contributing conditions like cardiovascular disease and drug use around the time of treatment. People with a history of heart attack, heart murmurs, arrhythmia, heart operation, or severe obesity are advised against taking ibogaine.
In the future, however, it may be possible to experience the positive effects of ibogaine in treating severe opioid addiction without the patient having to deal with side effects. Savant HWP, a pharmaceutical company in California, has developed a drug called 18-MC, which is chemically similar to ibogaine.
According to Scientific American, in 2014, the company completed phase I trials in Brazil with 14 healthy volunteers to determine whether the drug is safe, which they do not plan to publish. Savant HWP’s CEO was quoted saying the company’s formulation was “well tolerated” and there were “no serious adverse effects,” although it was “much more potent than we were expecting.” The drug has now entered phase II clinical trials in California, and if the results are promising, addiction treatment could be drastically changed.
Was this helpful?