US researchers demonstrated an implantable device that can deliver a drug payload when triggered by a remote wireless signal. After the payload is delivered, the whole system, including electronics, dissolve within weeks without harming the body in any way. Doctors would leave behind the resorbable packaging and electronics post surgery for infection management by either thermal treatment or by releasing an antibiotic. This way, there’s no need for oh-so dreaded followup surgeries. It also makes for an excellent solution when tackling deep infections or when systemic antibiotic treatment alone is insufficient due to the emergence of antibiotic-resistant strains.

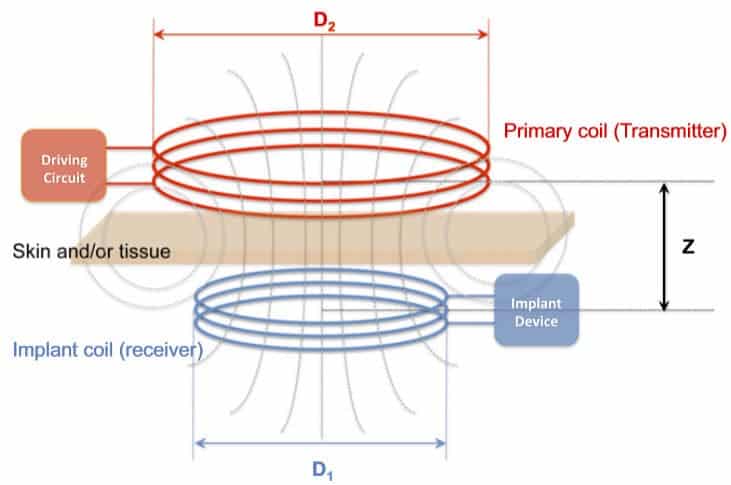
The main components of the implant are a resistor – used as a heat source for both releasing drugs and dissolving the implant – and a power-receiving coil made of magnesium deposited onto a silk protein”pocket” that also protects the electronics and controls its dissolution time.
The researchers from Tufts University and the University of Illinois at Champaign-Urbana implanted the devices in mice whose tissue was infected with Staphylococcus aureus. The implant was remotely ordered via a wireless signal to heat up and counter the infection with two sets of 10-minute sessions. Tissue was collected 24 hours later and no sign of infection was present in the infection site, nor in surrounding tissue. Devices completely dissolved after 15 days, and magnesium levels at the implant site and surrounding areas were comparable to levels typically found in the body.
A second round of experiments demonstrated the drug payload capabilities of the system. The implant was triggered to release the antibiotic ampicillin to kill E. coli and S. aureus bacteria which infected mouse tissue. The wireless activation of the devices was found to enhance antibiotic release without reducing antibiotic activity, according to the paper published in Proceedings of the National Academy of Sciences.
Previously, the same team demonstrated an imaging system that monitors tissue health from within a mouse, solar cells, as well as strain and temperature sensors – all capable of safely dissolving inside the body.
via KurweilAI
Was this helpful?



